Doubts and Solutions
OR
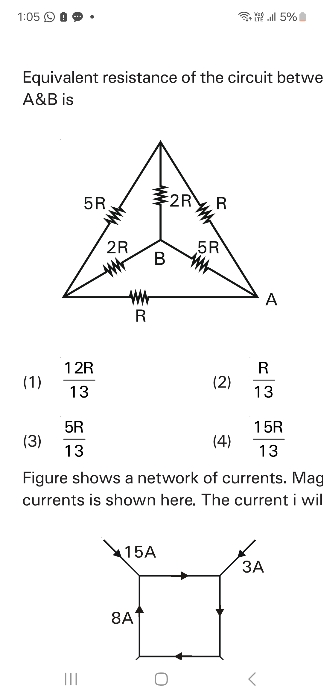
CBSE XI Science - Maths
Asked by tahikpreet0001 | 17 Apr, 2024, 06:41: AM
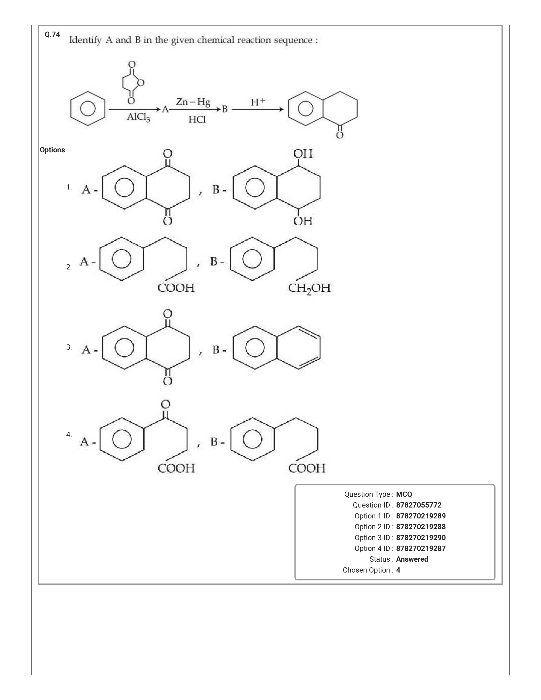
NEET NEET - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
CBSE VI - Grammar
Asked by beherajyoti253 | 16 Apr, 2024, 08:11: PM
CBSE XII Science - Maths
Asked by mahammadsharifdakhani67 | 16 Apr, 2024, 07:43: PM
CBSE VI - Social Studies
Asked by withmefun16 | 16 Apr, 2024, 07:21: PM