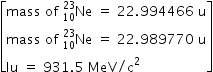CBSE Class 12-science Answered
During the formation of a nucleus, the protons and neutrons come closer to a distance of 10-14 m. The energy required for the purpose is spent by the nucleons at the expense of their masses. So mass of the nucleus found is less than the sum of the masses of the individual nucleons.
Since proton number and neutron number are conserved in a nuclear reaction, the total rest mass of neutrons and protons is the same on either side of a reaction. But the total binding energy of nuclei on the left side need not be the same as that on the right hand side. The difference in these binding energies appears as energy released or absorbed in a nuclear reaction.
OR
The graph showing the variation of binding energy per nucleon with mass number is shown below.
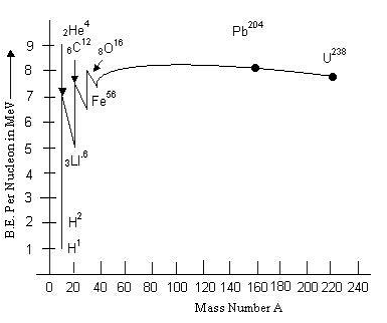
When we move from lighter nuclei to heavier nuclei, we find that there will be gain in the overall binding energy and hence release of energy. This indicates that energy can be released when two or more lighter nuclei fuse together to form a heavy nucleus. This process is called nuclear fusion.



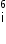 nucleus with subsequent emission of
nucleus with subsequent emission of 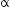 - particle Write the corresponding nuclear reaction. Calculate the energy released in this reaction.
Given mass of 3 L
- particle Write the corresponding nuclear reaction. Calculate the energy released in this reaction.
Given mass of 3 L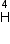 e = 4.000 26044 a.m.u.
Mass of neutron
e = 4.000 26044 a.m.u.
Mass of neutron 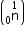 = 1.0086654 a.m.u.
Mass of tritium
= 1.0086654 a.m.u.
Mass of tritium 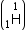 = 3.016049 a.m.u.
= 3.016049 a.m.u.
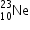 undergoes β- decay and becomes
undergoes β- decay and becomes 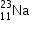 , Calculate the maximum kinetic energy of electrons emitted assuming that the daughter nucleus and anti- neutrino carry negligible kinetic energy.
, Calculate the maximum kinetic energy of electrons emitted assuming that the daughter nucleus and anti- neutrino carry negligible kinetic energy.