CBSE Class 12-science Answered
Why is it not possible to obtain a halide by treating ROH with 'X'?
Asked by sasikala106 | 08 Mar, 2018, 01:25: PM
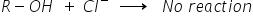
This is because in R-OH the bond between R and OH is a polar covalent bond which is a very strong bond.
And as a nucleophile Cl- is very weak than OH-. Hence it is not able to replace the strong R-OH bond with R-Cl
Hence to convert ROH into RCl we need a special reagent that is SOCl2 or PCl5, these reagents can bring this conversion.
Answered by Ramandeep | 08 Mar, 2018, 02:37: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by roshanisharma200611 | 07 Feb, 2024, 01:18: PM
CBSE 12-science - Chemistry
Asked by surekhas66675 | 02 Sep, 2021, 05:17: PM
CBSE 12-science - Chemistry
Asked by nazimb0313 | 02 Sep, 2020, 09:34: AM
CBSE 12-science - Chemistry
Asked by mastertask199 | 13 May, 2020, 04:08: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 03 Feb, 2020, 10:42: PM
CBSE 12-science - Chemistry
Asked by ajaysankhala051 | 11 Sep, 2019, 01:19: PM
CBSE 12-science - Chemistry
Asked by pragyachandraul03 | 23 Aug, 2019, 02:12: PM
CBSE 12-science - Chemistry
Asked by priadkonkar | 21 Jan, 2019, 08:52: PM
CBSE 12-science - Chemistry
Asked by rakeshraghav33 | 21 Jan, 2019, 02:49: PM
CBSE 12-science - Chemistry
Asked by Atulcaald | 16 May, 2018, 02:34: PM




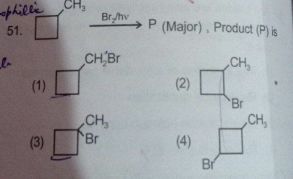
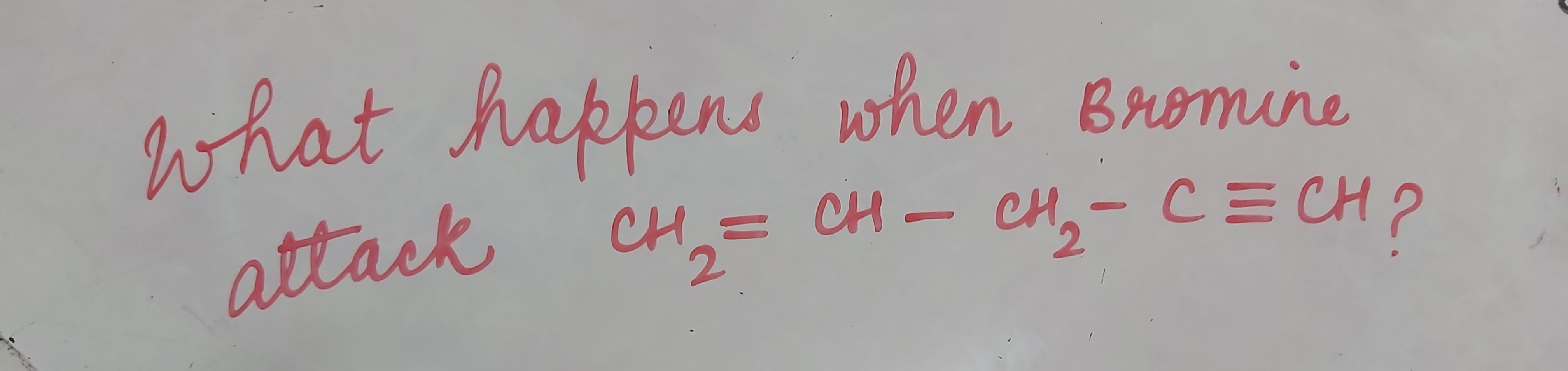
 Total No. of Mono Brominated product :-
Total No. of Mono Brominated product :-