CBSE Class 11-science Answered
Which postulates of kinetic theory are not valid at high pressure and low temperature? Give reasons to justify their invalidity?
Asked by Topperlearning User | 21 Apr, 2015, 01:06: PM
The postulates of the kinetic theory that are not valid at high pressure and low temperature are:
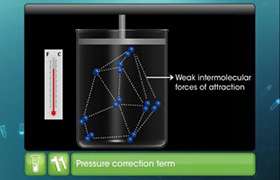
- There is no force of attraction between the molecules of a gas.
- Volume of the molecules of a gas is negligibly small in comparison to the space occupied by the gas.
According to the first assumption the gas will never liquefy but the gases do liquefy when cooled and compressed.
According to the second assumption the plot of P Vs V at constant temperature based on the experimental data and that based on theoretical calculations from Boyle’s law should coincide. But at high pressures deviations are observed which confirms that these postulates of the kinetic theory are invalid.
Answered by | 21 Apr, 2015, 03:06: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by khansuhana410 | 22 Dec, 2022, 11:48: AM
CBSE 11-science - Chemistry
Asked by aiqbal86592 | 28 May, 2019, 12:20: PM
CBSE 11-science - Chemistry
Asked by lovemaan5500 | 26 Jan, 2019, 12:57: PM
CBSE 11-science - Chemistry
Asked by nasankalagi04 | 17 Oct, 2018, 08:47: PM
CBSE 11-science - Chemistry
Asked by smanishkumar2002 | 04 Aug, 2018, 05:38: AM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 12:02: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 21 Apr, 2015, 12:19: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 21 Apr, 2015, 12:21: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 21 Apr, 2015, 12:31: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 17 Jun, 2016, 12:01: PM