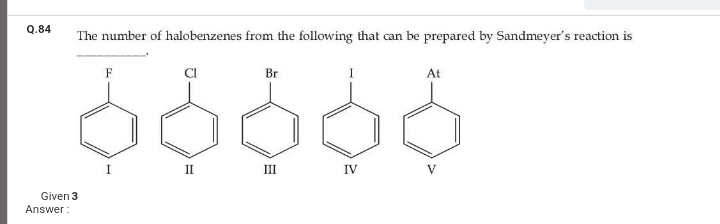
JEE Class main Answered
When a metal reacts with water it forms metal hydroxide
So does it get oxidised or does it get reduced?
Asked by atharvasunder | 19 Feb, 2021, 10:03: AM
Reaction of metal with water:
Metals react with water and produce a metal oxide and hydrogen gas. Metal oxides that are soluble in water dissolve in it to further form metal hydroxide. But all metals do not react with water.
Metal + Water → Metal oxide + Hydrogen
Metal oxide + Water → Metal hydroxide
Metal oxide + Water → Metal hydroxide
Product formation depends on the reactivity of the metals whether they will react with cold water, hot water, steam or will not react with water in any form at all.
More reactive metals like sodium and magnesium can react with simply water to form hydroxide because in solutions their hydroxides are stable (they are strongly basic in nature), while metals with lower reactivity (relatively lower reactive than sodium and magnesium) like iron react only with steam to first produce hydroxide which is not thermally stable and hence breaks down into iron oxide and water.
Thus, reactivity of metals, stability of products formed, rate of reaction, heat of reaction along with physical form of water are the responsible factors for the formation of oxide or hydroxide formation.
Hence above reaction is neither oxidation or reduction reaction.
But when metal reacts with oxygen that reaction ia called oxidation :
Metals react with oxygen to form corresponding oxides.
Metal + Oxygen → Metal oxide (Basic oxide)
Iron metal combines slowly with the oxygen of the air in the presence of water to form iron oxide. This iron oxide is the rust.
Oxidation of iron is called as rusting. Rusting involves slow oxidation of iron in presence of air and moisture.
4Fe + 3O2 → 2Fe2O3
Iron Oxygen Ferric oxide
2Fe2O3 + xH2O → Fe2O3.xH2O
(moisture) Rust
Hydrated iron (III)oxide
Metal + Oxygen → Metal oxide (Basic oxide)
Iron metal combines slowly with the oxygen of the air in the presence of water to form iron oxide. This iron oxide is the rust.
Oxidation of iron is called as rusting. Rusting involves slow oxidation of iron in presence of air and moisture.
4Fe + 3O2 → 2Fe2O3
Iron Oxygen Ferric oxide
2Fe2O3 + xH2O → Fe2O3.xH2O
(moisture) Rust
Hydrated iron (III)oxide
Answered by Ramandeep | 19 Feb, 2021, 12:13: PM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by muppanenicharitha | 14 Apr, 2024, 08:23: PM
JEE main - Chemistry
Asked by ruchisharmatbn | 06 Apr, 2024, 08:42: AM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 11:27: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by amarnathreddyp19 | 29 Mar, 2024, 06:47: AM
JEE main - Chemistry
Asked by syamalanandini49 | 19 Mar, 2024, 11:58: AM