CBSE Class 9 Answered
What is mole
Asked by Navrajdubeyjand | 19 Feb, 2019, 09:25: PM
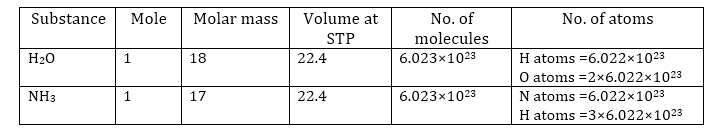
Mole concept-
1 mole of a substance is equal to its atomic mass or molecular mass expressed in grams.
The atomic mass of sodium is 23 grams.
Therefore, 23 grams of sodium is equal to one mole of sodium atoms.
Similarly, the molecular mass of oxygen (O2) = 2 × Atomic mass of oxygen
= 2 × 16 = 32 g
So, 32 grams of oxygen is equal to one mole of oxygen molecules.
1 mole (of anything) = 6.022 × 1023 in number
Answered by Varsha | 20 Feb, 2019, 10:53: AM
CBSE 9 - Chemistry
Asked by Navrajdubeyjand | 19 Feb, 2019, 09:25: PM

