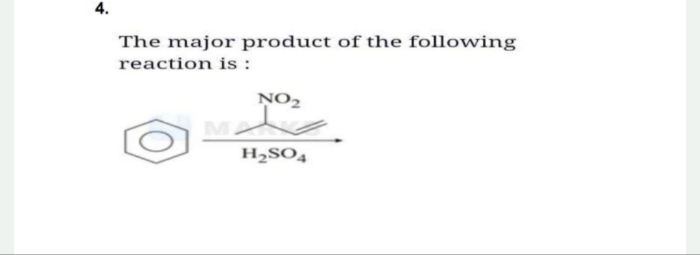
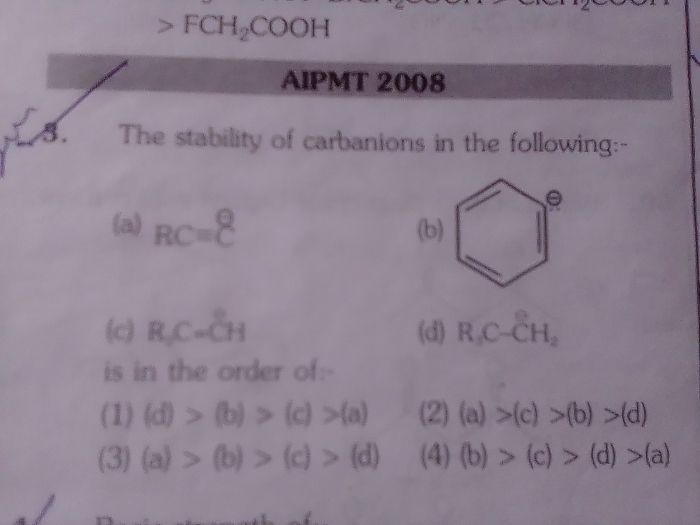
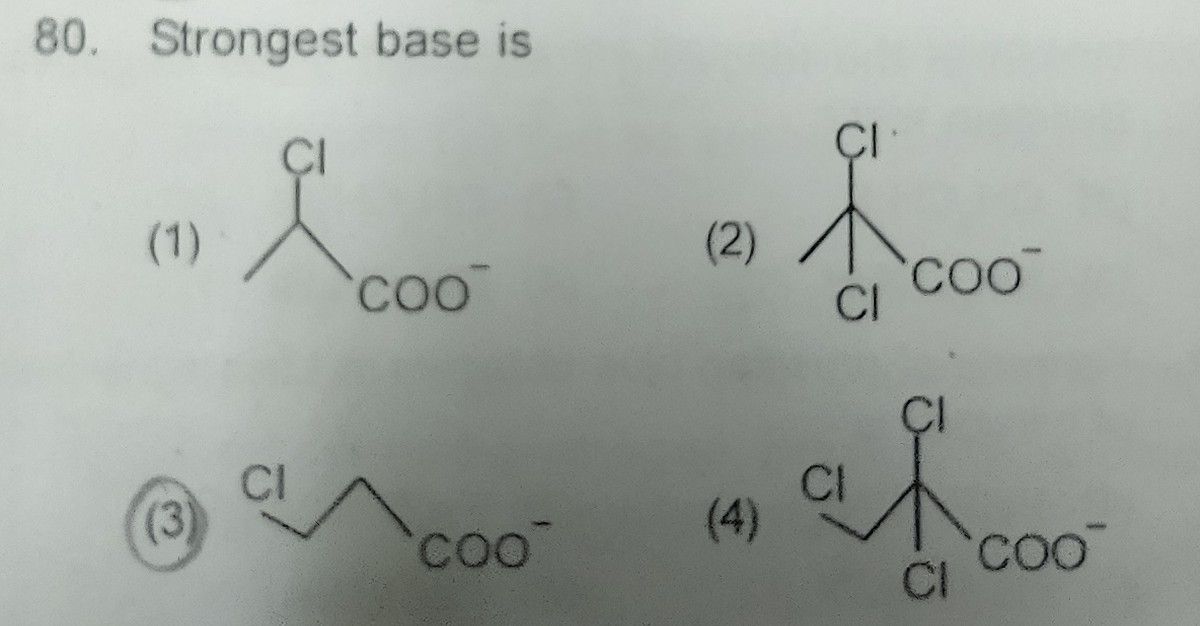
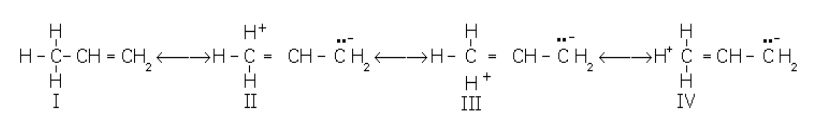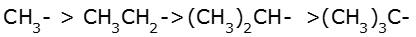NEET Class neet Answered
Hyperconjugation: It involves delocalisation of (s) electrons of C–H bond of an alkyl group directly attached to an atom of an unsaturated system or to an atom with an unshared p-orbital. The (s) electrons of C–H bond of the alkyl group enter into partial conjugation with the attached unsaturated system or with the unshared p-orbital. The interaction between the electrons of p systems (multiple bonds) and adjacent s bonds (single H–C bonds) of the substituent groups in organic compounds is called hypercojugation. It is a permanent effect.
Example: Hypercojugation in propene
Because there is no bond between the a-carbon atom and one of the hydrogen atoms, hyperconjugation is also called no-bond resonance. Although a free proton has been shown in the above structures, it is still bound quite firmly to the p-cloud, and hence it is not free to move.
Order of hyperconjugation: