CBSE Class 11-science Answered
Two p-orbitals from one atom and two p-orbitals from other atom are combined to form molecular orbitals. How many molecular orbitals will result from this combination? Explain.
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
Four molecular orbitals are formed when two p-orbitals from one atom combines with the two p-orbitals of the other atom. Out of these four molecular orbitals, two are bonding molecular orbitals and the other two are antibonding molecular orbitals.
Answered by | 04 Jun, 2014, 03:23: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by thesouro007 | 20 Mar, 2024, 06:05: AM
CBSE 11-science - Chemistry
Asked by shabnamaijaz83 | 19 Jun, 2022, 10:08: AM
CBSE 11-science - Chemistry
Asked by abnarsale | 31 Dec, 2021, 10:41: AM
CBSE 11-science - Chemistry
Asked by defence | 17 Feb, 2020, 05:09: PM
CBSE 11-science - Chemistry
Asked by mandriosa67 | 13 Feb, 2020, 03:03: PM
CBSE 11-science - Chemistry
Asked by Shrivatsa | 25 Aug, 2019, 02:11: PM
CBSE 11-science - Chemistry
Asked by sonkarshiva009 | 13 Mar, 2019, 05:47: PM
CBSE 11-science - Chemistry
Asked by vermaarti729 | 28 Feb, 2019, 08:28: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM



 4.IF7
5.
4.IF7
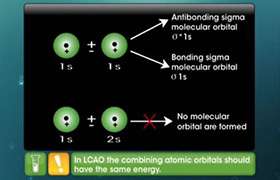
5. B.Explain anti bonding and bonding hybridised orbitals.
C._________on hydrolysis gives ethyne while ______ on hydrolysis gives methane.
D.Explain why the colour of Bayer's reagent gets discharged when treated with an alkene.
E.i) State and explain Le Chatelier’s principle. On the basis of this principle discuss the conditions for obtaining the maximum yield of SO3 in the following reaction. 2SO2(g)+ O2(g)⇌2SO3(g); ∆𝐻= - 42k.cal.(ii) Calculate the pH value of 0.01M CH3 COOH if it is 5% dissociated.
B.Explain anti bonding and bonding hybridised orbitals.
C._________on hydrolysis gives ethyne while ______ on hydrolysis gives methane.
D.Explain why the colour of Bayer's reagent gets discharged when treated with an alkene.
E.i) State and explain Le Chatelier’s principle. On the basis of this principle discuss the conditions for obtaining the maximum yield of SO3 in the following reaction. 2SO2(g)+ O2(g)⇌2SO3(g); ∆𝐻= - 42k.cal.(ii) Calculate the pH value of 0.01M CH3 COOH if it is 5% dissociated.