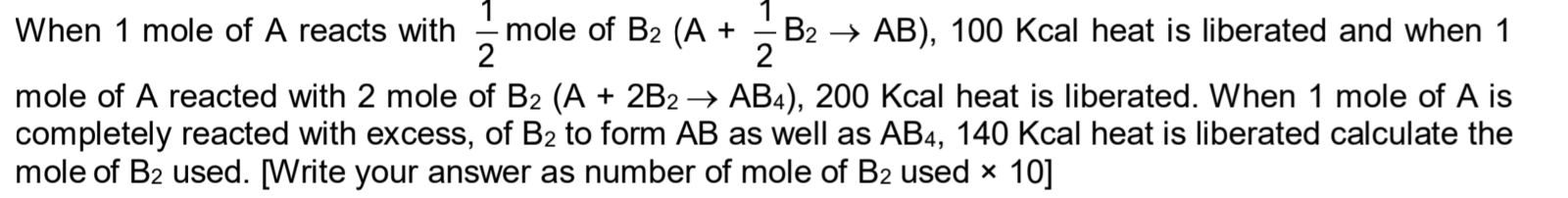CBSE Class 11-science Answered
The specific heat capacity of metal is 1 J/g/K . If equivalent mass of metal is 9 , calculate it's exact atomic mass
Asked by arunavamitra50 | 06 May, 2018, 08:11: PM
Given:
Specific heat = 1 J/gK
= 0.22 cal/g/mol
Equivalent weight = 9
By using Dulong Petit law,
Approximate atomic mass = 6.4 / Specific heat
=6.4/ 0.22
= 29.09 g
Valency = Approximate atomic mass / Equivalent weight
= 29.09 / 9
= 3.23
=3
Exact atomic weight = Equivalent weight × Valency
= 9× 3
= 27
Exact atomic weight of metal is 27 g.
Answered by Varsha | 07 May, 2018, 09:53: AM
Concept Videos
CBSE 11-science - Chemistry
Asked by drhimasingh | 22 May, 2020, 11:39: AM
CBSE 11-science - Chemistry
Asked by nareshrajpurohit43109 | 22 May, 2020, 11:18: AM
CBSE 11-science - Chemistry
Asked by d6knx7qmw1 | 15 May, 2020, 10:37: PM
CBSE 11-science - Chemistry
Asked by sahadipa1975 | 02 May, 2020, 08:53: AM
CBSE 11-science - Chemistry
Asked by abhishek19362771 | 08 Apr, 2020, 03:48: PM
CBSE 11-science - Chemistry
Asked by anilsolanki2060 | 22 Feb, 2020, 10:12: AM
CBSE 11-science - Chemistry
Asked by pujakurmi22 | 11 Nov, 2019, 10:59: PM
CBSE 11-science - Chemistry
Asked by jkatwara | 14 Oct, 2019, 12:21: PM
CBSE 11-science - Chemistry
Asked by vikas.kochhar6 | 30 Aug, 2019, 03:58: PM
CBSE 11-science - Chemistry
Asked by pb_ckt | 19 May, 2019, 11:56: PM