CBSE Class 9 Answered
Sulphur dioxide (SO2) is a colourless pungent smelling gas and is a major air pollutant.
(a) Write the electronic configuration of its constituent elements: Sulphur and Oxygen.
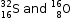 (b) Write valency of Sulphur and Oxygen.
(c) Are sulphur and oxygen isotopes of same element? Explain your answer.
(b) Write valency of Sulphur and Oxygen.
(c) Are sulphur and oxygen isotopes of same element? Explain your answer.
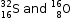 (b) Write valency of Sulphur and Oxygen.
(c) Are sulphur and oxygen isotopes of same element? Explain your answer.
(b) Write valency of Sulphur and Oxygen.
(c) Are sulphur and oxygen isotopes of same element? Explain your answer.
Asked by Topperlearning User | 04 Apr, 2016, 10:33: AM
(a) Electronic configuration of Sulphur (16) = 2, 8, 6
Electronic configuration of Oxygen (8) = 2, 6
(b) Valency of Sulphur and Oxygen = 2
(c) No, they are not isotopes as their atomic numbers are different.
Answered by | 04 Apr, 2016, 12:33: PM
Application Videos
Concept Videos
CBSE 9 - Chemistry
Asked by namyairmalwar9803.5sdatl | 07 Jun, 2020, 01:47: PM
CBSE 9 - Chemistry
Asked by pradoshsanesar | 10 Jan, 2020, 12:01: PM
CBSE 9 - Chemistry
Asked by maltidevi8800 | 06 Dec, 2019, 09:29: PM
CBSE 9 - Chemistry
Asked by ABHILASHA | 22 Aug, 2019, 08:46: AM
CBSE 9 - Chemistry
Asked by dubeykrishna49025 | 14 Oct, 2018, 10:02: PM
CBSE 9 - Chemistry
Asked by Topperlearning User | 13 May, 2014, 03:03: PM
CBSE 9 - Chemistry
Asked by Topperlearning User | 13 May, 2014, 03:03: PM
CBSE 9 - Chemistry
Asked by Topperlearning User | 13 May, 2014, 03:04: PM
CBSE 9 - Chemistry
Asked by Topperlearning User | 13 May, 2014, 03:04: PM
CBSE 9 - Chemistry
Asked by Topperlearning User | 13 May, 2014, 03:05: PM









