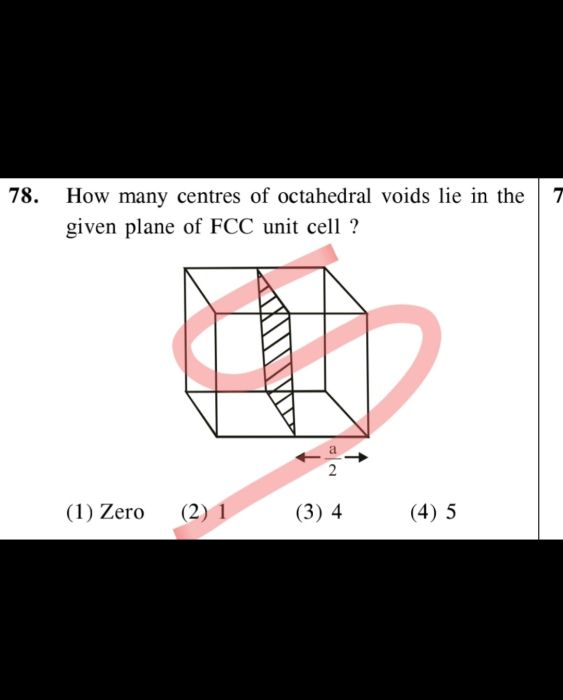
CBSE Class 12-science Answered
STAE DIFFERENCE BETWEEN THE TETRAHEDRAL AND OCTAHEDRAL VOIDS , THEIR LOCATIONS WITH THE HELP OF FIGURE
Asked by pardeepkumar2281 | 26 Sep, 2018, 09:01: PM
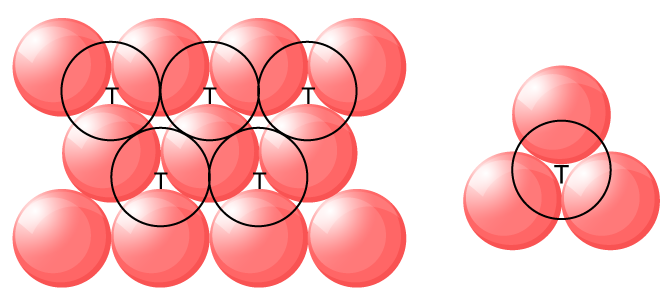
Tetrahedral voids:
The void surrounded by four spheres sitting at the corners of a regular tetrahedron is called a tetrahedral void.
Whenever the sphere of the second layer is above the void of the first layer a tetrahedral void is formed.
These voids are called tetrahedral voids because a tetrahedron is formed when these four spheres are joined.
Octahedral Voids:
The void surrounded by six
sitting at the corners of a regular octahedron is called an octahedral void.
The triangular voids in secnd layer are just above the triangular voids in the first layer.
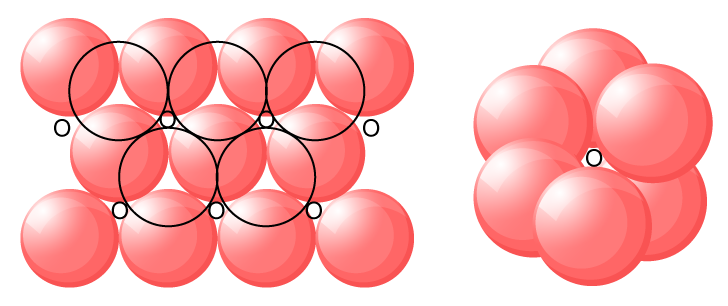
A simple triangular void ‘c’ which is surrounded by four spheres is called as tetrahedral void.
The double triangular void like 'd’ which is surrounded by six spheres is called as octahedral void.

Answered by Varsha | 28 Sep, 2018, 04:23: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by harshul2019 | 25 May, 2022, 08:52: PM
CBSE 12-science - Chemistry
Asked by arushidabhade | 17 Mar, 2021, 01:24: PM
CBSE 12-science - Chemistry
Asked by kasthurikalvi | 16 Sep, 2020, 03:46: PM
CBSE 12-science - Chemistry
Asked by sulaikhasulu393 | 01 Jul, 2020, 10:08: PM
CBSE 12-science - Chemistry
Asked by anukritisingh8103.bmps | 18 May, 2020, 03:00: PM
CBSE 12-science - Chemistry
Asked by rohitraman1115 | 10 Sep, 2019, 07:10: PM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 05 Aug, 2019, 12:08: AM
CBSE 12-science - Chemistry
Asked by prakriti12oct | 04 Aug, 2019, 09:06: PM
CBSE 12-science - Chemistry
Asked by ranasingh04082002 | 31 Jul, 2019, 02:04: PM
CBSE 12-science - Chemistry
Asked by Saransekar407 | 11 Mar, 2019, 06:51: PM