Someone pls do this
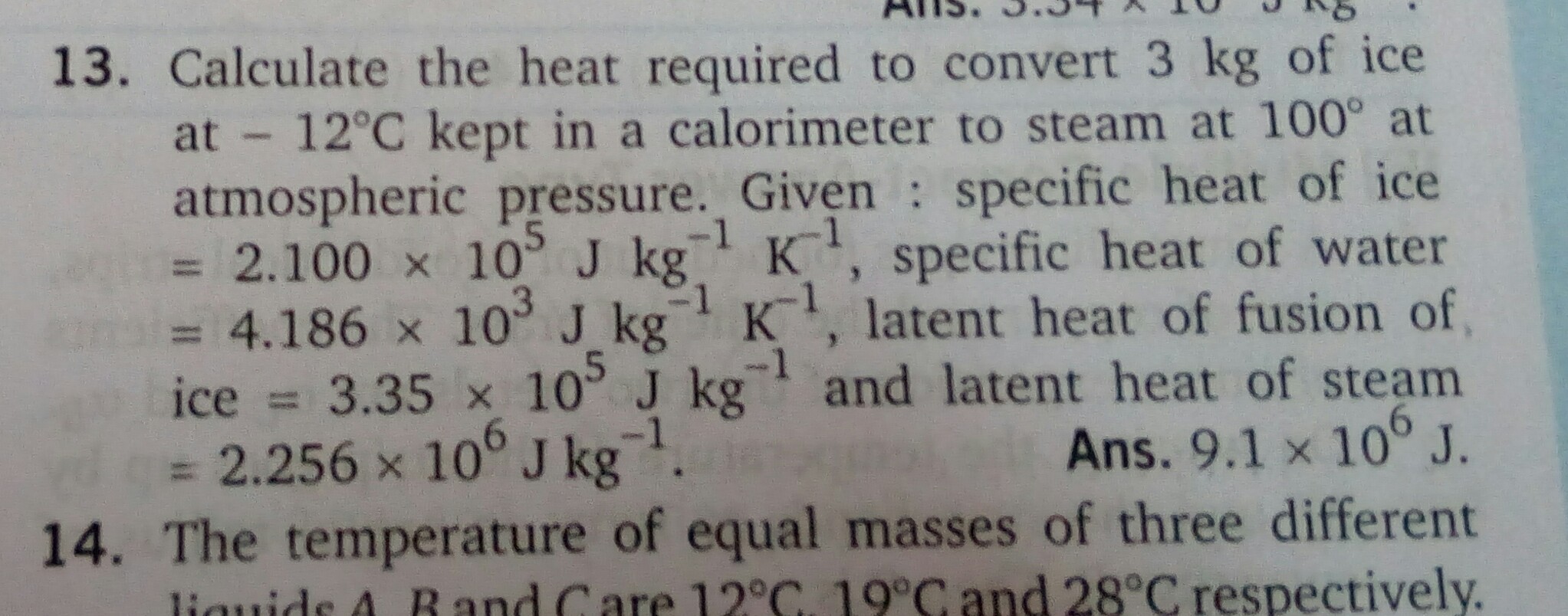
- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- 9
- 10
You have rated this answer /10
Browse free questions and answers by Chapters
- 1 Units and Measurement
- 2 Motion in a Straight Line
- 3 Motion in a Plane
- 4 Laws of Motion
- 5 Work, Energy and Power
- 6 System of Particles and Rotational Motion
- 7 Gravitation
- 8 Mechanical Properties of Solids
- 9 Mechanical Properties of Fluids
- 10 Thermal Properties of Matter
- 11 Thermodynamics
- 12 Kinetic Theory
- 13 Oscillations
- 14 Waves
- 15 Physical World
