ICSE Class 10 Answered
- Electrolysis of Fused Lead Bromide
Electrolyte: Molten lead bromide (PbBr2)
Electrodes:
Anode: Graphite
Cathode: Graphite
Ions present:
Pb2+ and Br–
Overall reaction: PbBr2 → Pb2+ + 2Br–
Electrode reactions:
Reaction at the cathode: Pb2+ + 2e− → Pb
Reaction at the anode: Br– − e− → Br
Br + Br → Br2
- Electrolysis of Acidified Water
Electrodes:
Anode: Platinum
Cathode: Platinum
Ionisation of acidified water: H2O
Electrodes:
Anode: Platinum
Cathode: Platinum
Ionisation of acidified water: 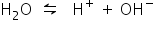
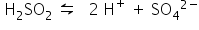
Ions present: H+, SO42–, OH–
Electrode reactions:
Reaction at the cathode: H+ + e− → H
H + H → H2 (hydrogen molecule)
Reaction at the anode: OH– − e− → OH × 4
OH– ion discharge in preference to SO42–
4OH → 2H2O + O2 (oxygen molecule)






 is the oxide of a metal 'M' which is above hydrogen in electrochemical series.
is the oxide of a metal 'M' which is above hydrogen in electrochemical series.