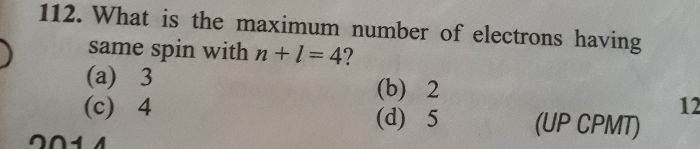CBSE Class 11-science Answered
Q)

Asked by araima2001 | 09 Mar, 2017, 07:56: AM
Option (c)
NO2 → N2O4
The above reaction is a dimerisation reaction.
NO2 is paramagnetic since in NO2 we have:
One N=O
One N- O
And one unpaired electron on the N atom.
After the dimerisation reaction, N2O4 formed is diamagnetic since the unpaired electron gets paired.
Hence there is decrease in paramagnetism.
Answered by Vaibhav Chavan | 10 Mar, 2017, 12:30: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by deba.biswas561 | 14 Jun, 2022, 08:07: AM
CBSE 11-science - Chemistry
Asked by Neetukhaiwal | 24 Jul, 2021, 11:18: AM
CBSE 11-science - Chemistry
Asked by salvadormiranda2509 | 13 Feb, 2021, 12:53: PM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 01 May, 2020, 04:37: PM
CBSE 11-science - Chemistry
Asked by prashantyadav592 | 29 Jan, 2020, 09:39: AM
CBSE 11-science - Chemistry
Asked by Debdulal | 29 Aug, 2019, 04:47: PM
CBSE 11-science - Chemistry
Asked by sairamsribaba80 | 28 Aug, 2019, 06:54: PM
CBSE 11-science - Chemistry
Asked by timeyashkadam | 19 Apr, 2019, 10:22: AM
CBSE 11-science - Chemistry
Asked by himankarbhati3 | 21 Sep, 2018, 08:29: AM
CBSE 11-science - Chemistry
Asked by ramandeep.kaur | 06 Sep, 2018, 01:21: PM