NEET Class neet Answered
Pls solving the follin

Asked by ntg432000 | 28 Apr, 2019, 04:25: PM
Q. 79.
Given:
Mass = 15 g
Volume = 500 ml
We know,
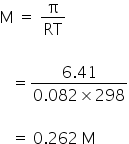
Therefore, 0.262 mol of particles present in 1 litre of solution and 0.131 mol of particles present in 500 ml of solution.
Each mole of sucrose gives 1 mol of particles.
Each mole of NaCl gives 2moles of particals.
Consider,
Mass of C12H22O11 be X g and
Mass of NaCl is (15 - X) g

Pertange composition,
Sucrose is 81.42 %
NaCl is 18.57%
Answered by Varsha | 29 Apr, 2019, 02:33: PM
Concept Videos
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by ramadevisupriya5678 | 28 Mar, 2024, 02:18: PM
NEET neet - Chemistry
Asked by myindiaisbad | 17 Jun, 2022, 11:17: AM
NEET neet - Chemistry
Asked by bhaveshkaria31 | 30 May, 2022, 09:26: PM
NEET neet - Chemistry
Asked by rautganesh2255 | 01 Jul, 2021, 09:32: AM
NEET neet - Chemistry
Asked by NituBarman192 | 01 Jun, 2021, 10:22: PM
NEET neet - Chemistry
Asked by bhagirathdangi12345 | 12 Feb, 2021, 01:42: PM
NEET neet - Chemistry
Asked by akashmanu09 | 08 Jan, 2021, 10:21: AM
NEET neet - Chemistry
Asked by arnavvidudala20050 | 17 May, 2020, 03:07: PM