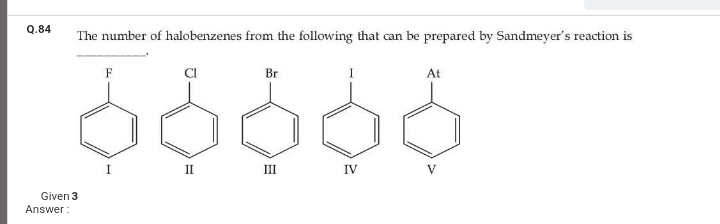
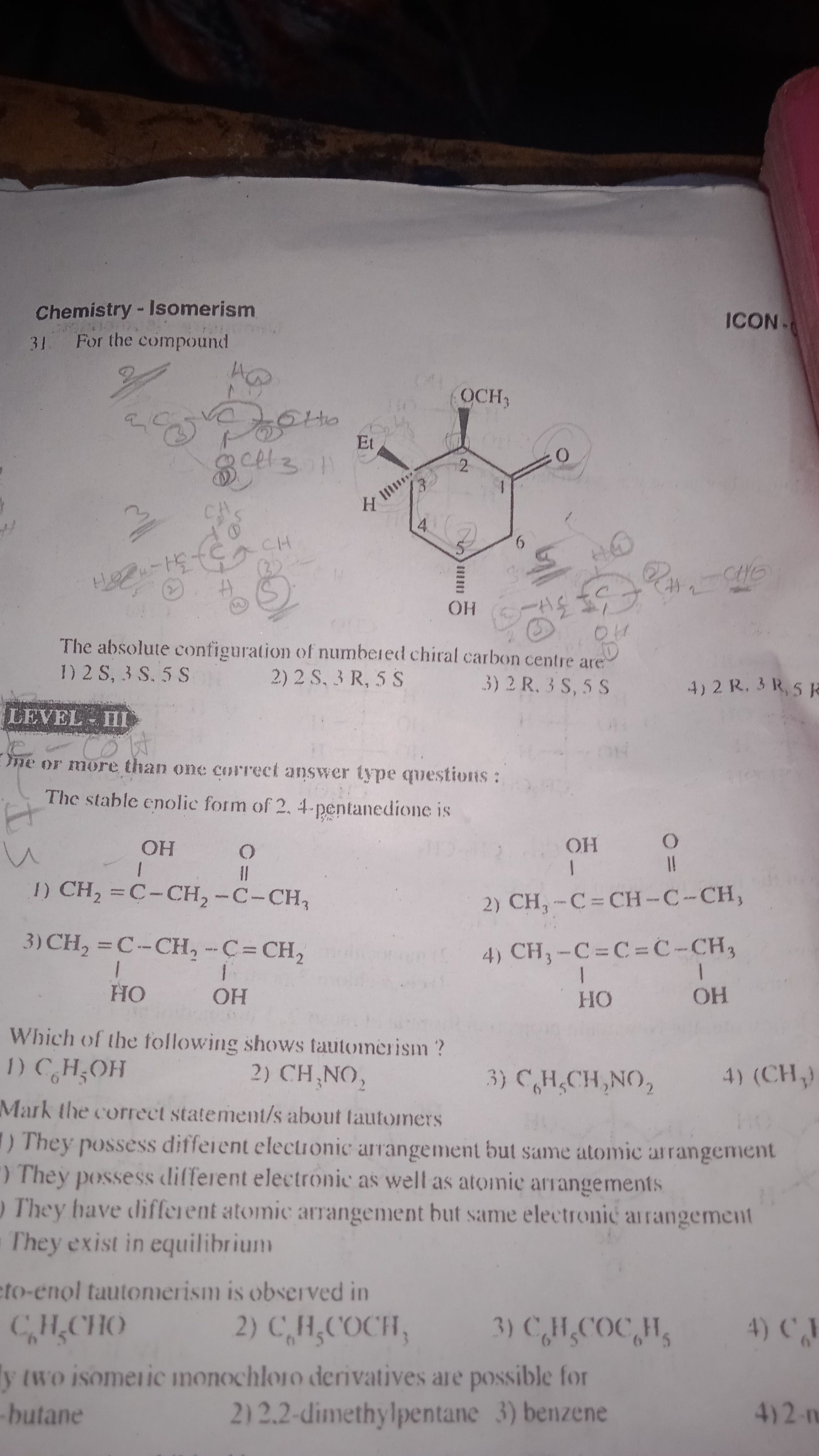
JEE Class main Answered
Please explain the following
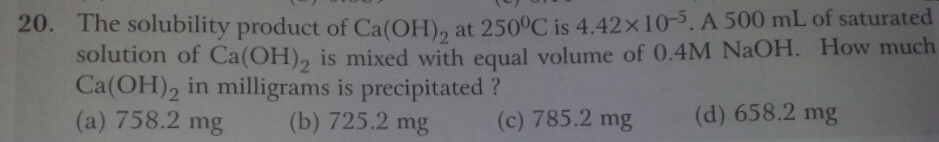
Asked by Balbir | 24 Dec, 2018, 08:09: AM
Let the solubility of Ca(OH)2 be 'x' mol/lit

Amount of Ca(OH)2 precipitated = 742.1 mg.
Answered by Varsha | 24 Dec, 2018, 01:18: PM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by muppanenicharitha | 14 Apr, 2024, 08:23: PM
JEE main - Chemistry
Asked by ruchisharmatbn | 06 Apr, 2024, 08:42: AM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 11:27: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by amarnathreddyp19 | 29 Mar, 2024, 06:47: AM
JEE main - Chemistry
Asked by syamalanandini49 | 19 Mar, 2024, 11:58: AM