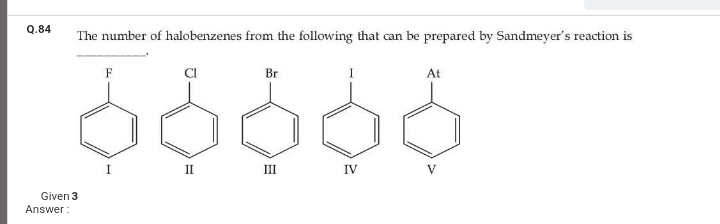
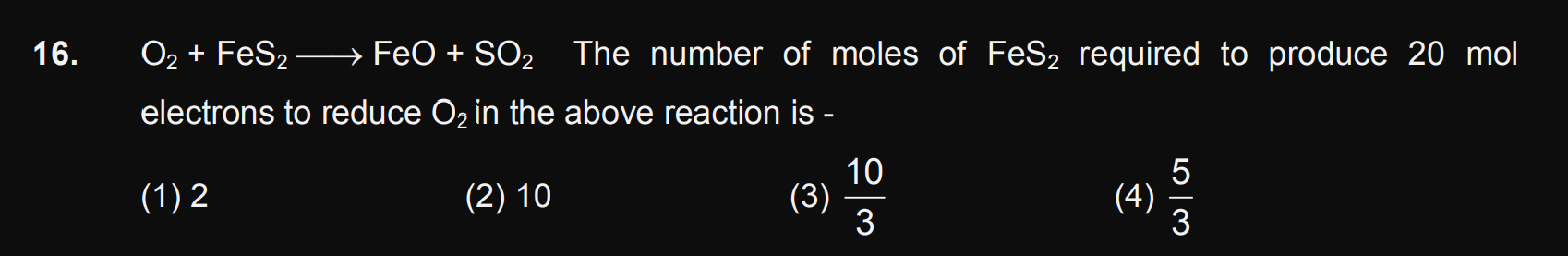JEE Class main Answered
Please expalin the following

Asked by Balbir | 23 Dec, 2018, 03:50: PM
MgCl2 + 2NaOH → Mg(OH)2 + 2NaCl
No of Millimoles (t0) 10 20 0 0
No of Millimoles (t) 0 0 10
So, [Mg2+] = 10/200 = 1/20
Ksp Mg(OH)2 = S[Mg2+] × S(OH-) = {1/20} × {4 (OH-)2}
2 (OH-) = 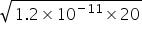 =
= 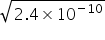 =
= 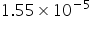
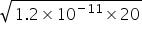 =
= 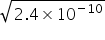 =
= 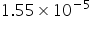
(OH-) = Near to 10-5
Answered by Sumit Chakrapani | 03 Jan, 2019, 03:56: AM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 01:21: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 09:44: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM
JEE main - Chemistry
Asked by muppanenicharitha | 14 Apr, 2024, 08:23: PM
JEE main - Chemistry
Asked by ruchisharmatbn | 06 Apr, 2024, 08:42: AM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 11:27: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by amarnathreddyp19 | 29 Mar, 2024, 06:47: AM
JEE main - Chemistry
Asked by syamalanandini49 | 19 Mar, 2024, 11:58: AM