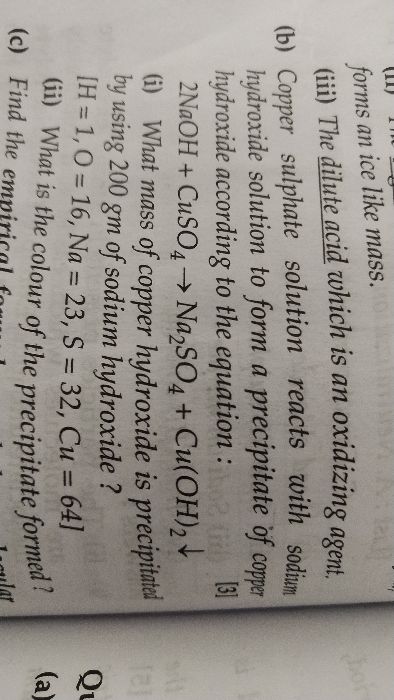ICSE Class 10 Answered
Please Answer with reference to Gay Lussac's Law:-
10 litres of propane[C3H8][60%] and Butane[C4H10][40%] is burnt. Calculate the total volume of carbon dioxide formed. Combustion reactions of the mixture are represented as -
C3H8 + 5O2 -----> 3CO2 + 4H2O
2C4H10 + 13O2 --------> 8CO2 + 10H2O
Asked by vprincesolomon07 | 07 Aug, 2018, 11:12: PM
C3H8 + 5O2 → 3CO2 + 4H2O
2C4H10 + 13O2 → 8CO2 + 10H2O
60 ml of propane (C3H8) gives 3 X 60 = 180 ml CO2
40 ml of butane (C4H10) gives = 8 X 40/2 = 160 ml of CO2
Total carbon dioxide produced = 340 ml
So, when 10 litres of the mixture is burnt = 34 litres of CO2 is produced.
Answered by Ramandeep | 08 Aug, 2018, 11:11: AM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by jrvedant208 | 05 Feb, 2024, 10:37: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 10 Jul, 2022, 10:13: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 25 Jun, 2022, 10:24: PM
ICSE 10 - Chemistry
Asked by palshivom72 | 14 Jul, 2020, 07:56: PM
ICSE 10 - Chemistry
Asked by jhabijay01 | 27 May, 2020, 12:20: PM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:53: AM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:37: AM
ICSE 10 - Chemistry
Asked by aashimegh | 28 Aug, 2019, 05:25: PM