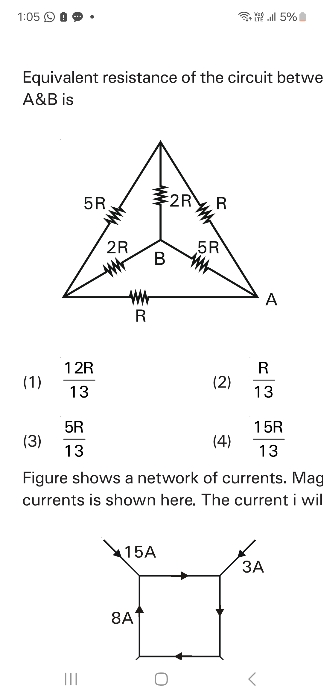
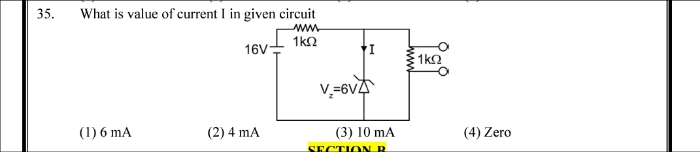
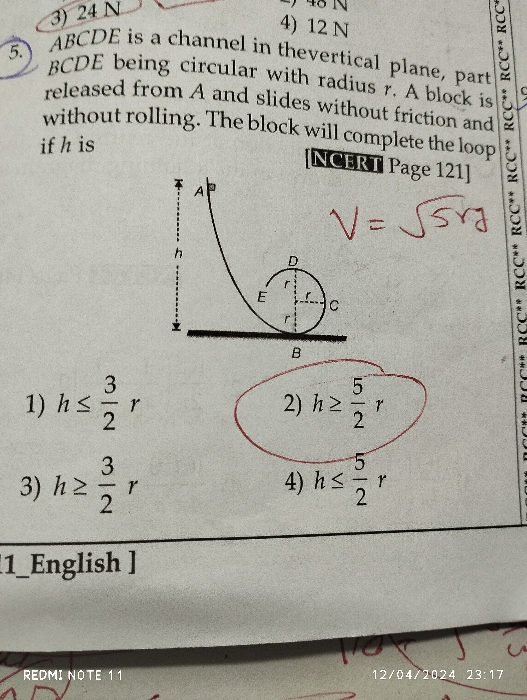NEET Class neet Answered
please answer this
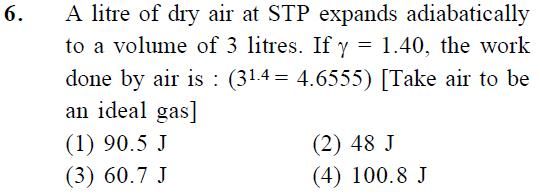
Asked by Prashant DIGHE | 29 Jan, 2020, 10:22: PM
Workdone by adiabatic process W is given by
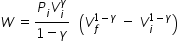
where Pi is initial pressure, Vi is initial volume, Vf is final volume and γ is adiabatic constant
we have initial pressure at STP, Pi = 1.013 × 105 Pa , initial volume Vi = 1 litre = 10-3 m3
and final volume Vf = 3 litre = 3 × 10-3 m3
Workdone W = [ 1.013 × 105 × (10-3 )γ / (-0.4) ] × [ 3-0.4 - 1 ] × (10-3)1-γ
W = [ 101.3 / (-0.4)] ×[ 3-0.4 - 1 ] = 90 J
Answered by Thiyagarajan K | 30 Jan, 2020, 11:09: AM
Application Videos
NEET neet - Physics
Asked by ramanjaneyuluoguru | 25 Apr, 2024, 04:18: PM
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM