CBSE XI Science Chemistry Redox Reactions
sir,here is my question on oxidation number
NEET1 NEET1 Chemistry Coordination Compounds
Q.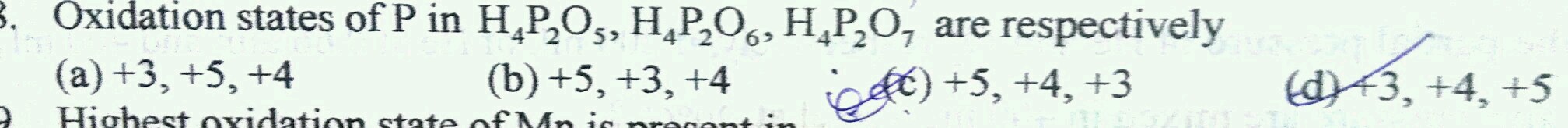
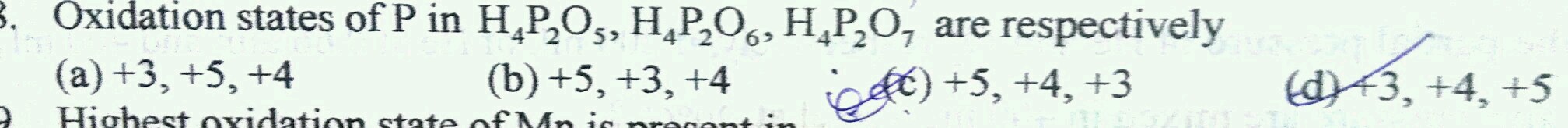
NEET1 NEET1 Chemistry Stoichiometry and Redox Reactions
Q.

JEE Plances JEE (Main) Chemistry Co-ordination Compounds
Q.

CBSE XI Science Chemistry Some Basic Concepts of Chemistry
what is oxidation state and how do you find it?
JEE Plances JEE (Main) Chemistry
Could you just share the link of the video lecture of oxidation number?
CBSE XII Science Chemistry The d-Block and f-Block Elements
1) which one of l and
and  is not likely to exist ?
is not likely to exist ?
2) how does ammonia react with AgN ? Write it chemical reaction ?
? Write it chemical reaction ?
3) give the disproportionation reaction of 
4) HYPOPHOSPHURUS acis is a good reducing agent . Justify with example
5) give chemical equation in support of the statement that all bonds in P MOLECULE ARE NOT EQUIVALENT
MOLECULE ARE NOT EQUIVALENT
 and
and  is not likely to exist ?
is not likely to exist ? ? Write it chemical reaction ?
? Write it chemical reaction ?
 MOLECULE ARE NOT EQUIVALENT
MOLECULE ARE NOT EQUIVALENT CBSE XI Science Chemistry Redox Reactions
Give the oxidation number of all elements in CH3COOH? In my book the oxidation number of oxygen in this compound is -1? How
CBSE XII Science Chemistry Coordination Compounds
Find the difference in the oxidation numbernumbers present of the two types of sulphur present in Na2S4O6 [sodium - 2 sulphur - 4 oxygen - 6]. (Hint :- sulphur-sulphur linkages present)
CBSE XI Science Chemistry Redox Reactions and Electrochemistry
Plz solve it step by step
CBSE XI Science Chemistry Some Basic Concepts of Chemistry
Pls solve.
NEET NEET Chemistry Some Basic Concepts in Chemistry
Equivalent weight is
CBSE XII Science Chemistry The p-Block Elements
The incorrect order of decreasing oxidation number of S in compounds is: H2 S2 O7 > Na2 S4 O6 > Na2 S2 O3 > S8 H2 S O5 > H2 S O3 > S Cl2 > H2 S S O3 > S O2 > S8 > H2 S H2 S O4 > S O2 > H2 S > H2 S2 O8
CBSE XI Science Chemistry Redox Reactions
(i) Balance the following equations by oxidation number method:
(1) Cu + HNO3 → Cu (NO3)2 + NO2 + H2O)
(2) K2Cr2O7 + HCl → KCl + CrCl3 + H2O + Cl2
(ii) Give reasons for the following:
(1) HNO3 acts only as an oxidising agent while HNO2 can act both as a
reducing agent and an oxidising agent.
(2) Chlorine liberates iodine from KI solution.
How will you convert the following? (Give balanced equation.)
(1) Sodium acetate to methane
Find the oxidation number of:
(1) S in Na2S4 O6
2) S in 
(1) Cu + HNO3 → Cu (NO3)2 + NO2 + H2O)
(2) K2Cr2O7 + HCl → KCl + CrCl3 + H2O + Cl2
(ii) Give reasons for the following:
(1) HNO3 acts only as an oxidising agent while HNO2 can act both as a
reducing agent and an oxidising agent.
(2) Chlorine liberates iodine from KI solution.
(1) Sodium acetate to methane
(1) S in Na2S4 O6

