CBSE Class 11-science Answered
Mixture of the propane and butane occupy 3 liter volume complete combustion of this mixture produced 10 liters co^2 find the constitution of this mixture
Asked by mannkhatri112 | 01 May, 2019, 07:24: AM
The combustion reactions are as;
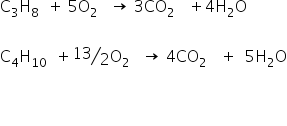
Let the volume of propane be x lit and volume of butane be 3-x lit.
CO2 formed by combustion of propane is 3x
CO2 formed by combustion of butane is 4(3-x)
We have,
x+y =10 lit
Therefore
3x + 4(3-x) = 10
3x + 12 -4x = 10
x = 2 lit
As x is volume of propane = 2 lit
and volume of butane = (3 - x)
=3-2 = 1 lit
Therefore the composition of mixture is
Propane : Butane is 2:1
Answered by Varsha | 02 May, 2019, 02:45: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by jayag1298 | 08 Apr, 2024, 03:14: PM
CBSE 11-science - Chemistry
Asked by omniscientnjf2021 | 07 Apr, 2024, 10:18: PM
CBSE 11-science - Chemistry
Asked by hcnainwal | 15 Jun, 2023, 10:39: AM
CBSE 11-science - Chemistry
Asked by Jprmumal29 | 18 Dec, 2022, 09:48: PM
CBSE 11-science - Chemistry
Asked by mallikarjunasangi28 | 22 Jul, 2022, 07:57: PM
CBSE 11-science - Chemistry
Asked by vedwatisharma79 | 10 Jun, 2022, 05:27: PM
CBSE 11-science - Chemistry
Asked by thathvakunjusree | 10 Dec, 2021, 06:46: AM
CBSE 11-science - Chemistry
Asked by udheshraddha2004 | 28 Oct, 2021, 09:37: PM
CBSE 11-science - Chemistry
Asked by arunparewa2000 | 27 Oct, 2021, 06:59: PM
CBSE 11-science - Chemistry
Asked by arttameher038 | 23 Aug, 2021, 07:06: AM







