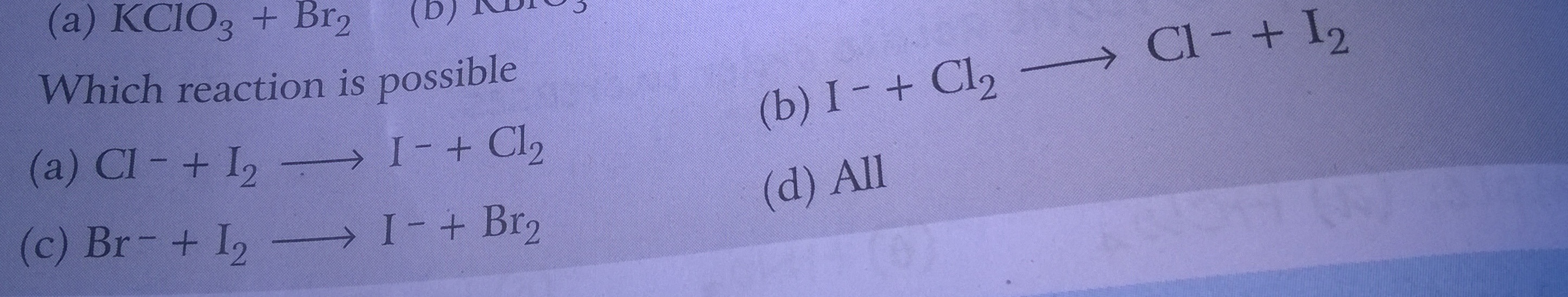CBSE Class 11-science Answered
Justify that the reaction 2Cu2O(s)+Cu2S(s) → 6Cu(s)+SO2(g) is a redox reaction.
Asked by Topperlearning User | 15 Apr, 2015, 11:14: AM
Writing the oxidation number of each atom above its symbol, we have
+1 -2 +1 -2 0 +4 -2
2Cu2O(s) + Cu2S(s) → 6Cu(s) + SO2(g)
Here in the reaction, the oxidation number of copper decreases from +1 in Cu2O or Cu2S to 0 in copper metal, therefore copper is reduced. Further the oxidation number of S increases from -2 in Cu2S to +4 in SO2, therefore S is oxidised. Therefore, this reaction is a redox reaction.
Answered by | 15 Apr, 2015, 01:14: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by jaip83491 | 24 Jan, 2021, 10:43: AM
CBSE 11-science - Chemistry
Asked by sulaikhasulu393 | 07 Jun, 2020, 10:51: PM
CBSE 11-science - Chemistry
Asked by debjit_dm | 04 May, 2020, 03:08: PM
CBSE 11-science - Chemistry
Asked by Balbir | 30 Jun, 2019, 03:45: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 05 Sep, 2018, 01:32: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 09 Aug, 2018, 04:52: PM
CBSE 11-science - Chemistry
Asked by dineshchem108 | 17 Jul, 2018, 06:02: PM
CBSE 11-science - Chemistry
Asked by vaagai2353 | 29 Jun, 2018, 09:55: PM
CBSE 11-science - Chemistry
Asked by Balbir | 19 Jun, 2018, 06:36: PM