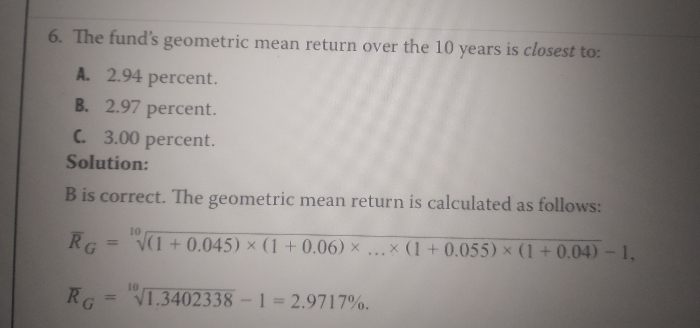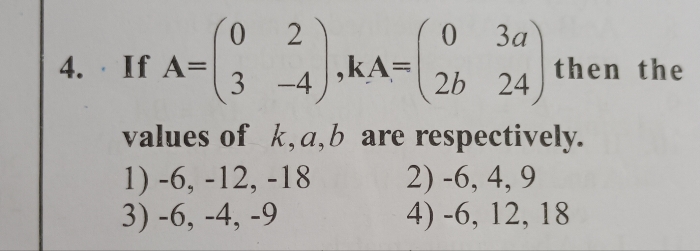JEE Main Questions and Answers
JEE Main - Maths
Asked by usakoyalshailesh | 15 Apr, 2024, 09:48: AM
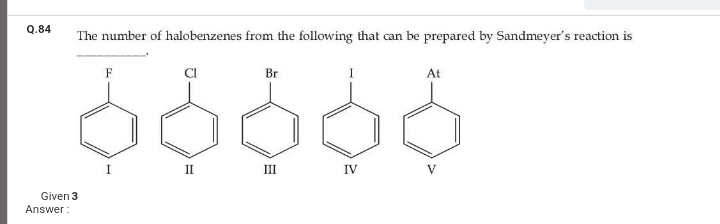
JEE Main - Chemistry
Asked by muppanenicharitha | 14 Apr, 2024, 08:23: PM
JEE Main - Maths
Asked by sarthakshukla7275 | 14 Apr, 2024, 01:29: PM
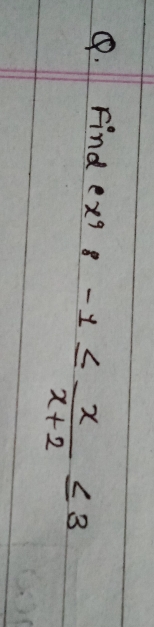
JEE Main - Maths
Asked by ashwinskrishna2006 | 14 Apr, 2024, 11:58: AM
JEE Main - Maths
Asked by satyampatel95694 | 13 Apr, 2024, 05:35: PM
JEE Main - Maths
Asked by ashwinskrishna2006 | 13 Apr, 2024, 11:25: AM
JEE Main - Maths
Asked by vidyanath.j | 11 Apr, 2024, 09:28: PM
JEE Main - Maths
Asked by rahulkumarbsc73 | 09 Apr, 2024, 04:33: PM
JEE Main - Maths
Asked by srinivaskarri79 | 08 Apr, 2024, 09:53: PM