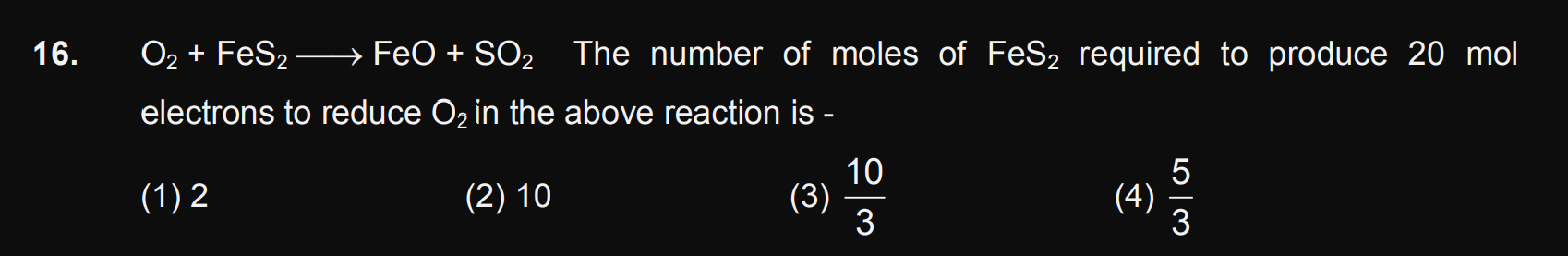JEE Main Questions and Answers
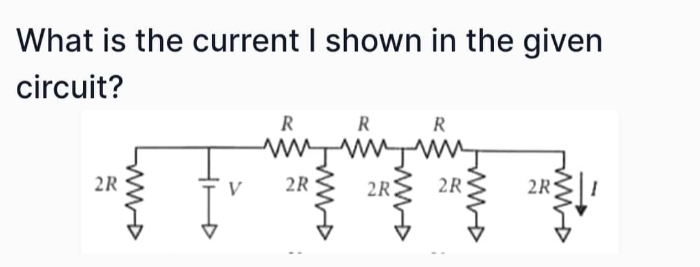
JEE Main - Physics
Asked by arivaryakashyap | 23 Apr, 2024, 10:40: AM
JEE Main - Physics
Asked by ratnadeep.dmr003 | 21 Apr, 2024, 11:06: PM
JEE Main - Maths
Asked by kakumanumanoj282 | 20 Apr, 2024, 06:07: PM
JEE Main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE Main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 01:21: PM
JEE Main - Physics
Asked by ksahu8511 | 19 Apr, 2024, 11:55: AM
JEE Main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 09:44: PM
JEE Main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM