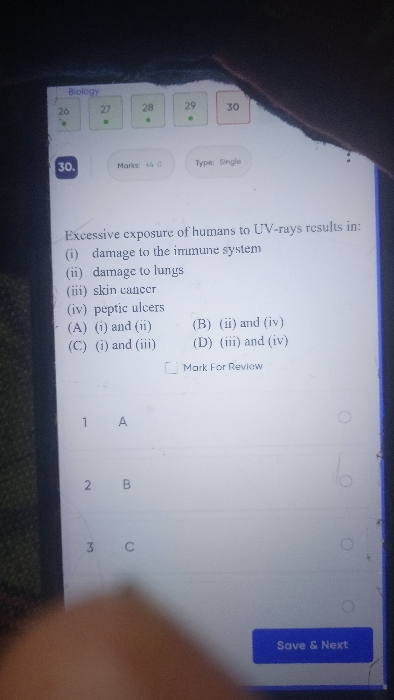
CBSE Class 10 Answered
In question 8th why we use alk KMnO4 instead of CrO3 for oxidation of alcohol
Asked by meenakshi2005suhag | 07 Jan, 2019, 08:02: PM
When ethanol reacts with chromium trioxide i.e., CrO3 (only partial oxidation occurs and ethanal (acetaldehyde) is formed.
While when ethanol is heated with alkaline KMnO4 it forms ethanoic acid (acetic acid) due to complete combustion.
Hence oxidation of ethanol with CrO3 produces ethanal while ethanol, when oxidised with alkaline KMnO4, produces ethanoic acid.
Answered by Ramandeep | 10 Jan, 2019, 12:00: PM
CBSE 10 - Science
Asked by sony.lucky455 | 02 Apr, 2024, 03:23: PM
CBSE 10 - Science
Asked by sagarmishra | 18 Mar, 2024, 04:10: PM
CBSE 10 - Science
Asked by srinivasansriharinia9 | 01 Mar, 2024, 08:17: PM
CBSE 10 - Science
Asked by muskanprayagraj78 | 26 Feb, 2024, 05:44: PM
CBSE 10 - Science
Asked by kmannatpreet4 | 21 Feb, 2024, 07:34: PM
CBSE 10 - Science
Asked by umaambiger87 | 08 Feb, 2024, 03:28: PM
CBSE 10 - Science
Asked by pvasanth3573 | 07 Feb, 2024, 10:13: PM
CBSE 10 - Science
Asked by samirankarmakar2702 | 07 Feb, 2024, 09:59: PM