JEE Class main Answered
In a reversible reaction 2NO2<--K1—K2---->N2O4, the rate of disappearance of NO2 is equal to:(K1 is rate of forward reaction, K2 rate of backward reaction.)
1) 2K1[NO2]2/K2
2) 2K1[NO2]2-2K2[N2O4]
3) 2K1[NO2]2-K2[N2O4]
4) 2(K1-K2)[NO2]
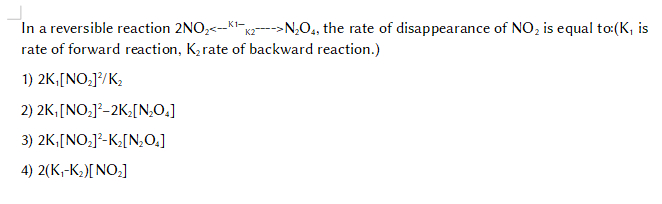
Asked by koushikr063 | 06 May, 2019, 10:37: AM
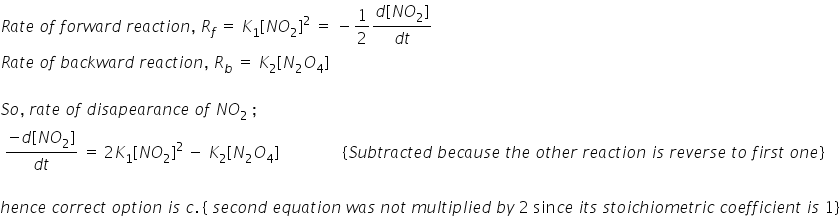
Answered by Sumit Chakrapani | 06 May, 2019, 11:21: AM
Application Videos
Concept Videos
JEE main - Chemistry
Asked by cheekatiyogendra143 | 20 Apr, 2024, 11:16: AM
JEE main - Chemistry
Asked by jwhhebbb | 19 Apr, 2024, 01:21: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 09:44: PM
JEE main - Chemistry
Asked by ashwinskrishna2006 | 18 Apr, 2024, 05:37: PM
JEE main - Chemistry
Asked by muppanenicharitha | 14 Apr, 2024, 08:23: PM
JEE main - Chemistry
Asked by ruchisharmatbn | 06 Apr, 2024, 08:42: AM
JEE main - Chemistry
Asked by adityadoodi3 | 05 Apr, 2024, 11:27: PM
JEE main - Chemistry
Asked by gmafia618 | 04 Apr, 2024, 08:48: PM
JEE main - Chemistry
Asked by amarnathreddyp19 | 29 Mar, 2024, 06:47: AM
JEE main - Chemistry
Asked by syamalanandini49 | 19 Mar, 2024, 11:58: AM