NEET Class neet Answered
If potential energy is taken to be zero for an electron in ground state of hydrogen atom, then calculate its total mechanical energy in the 1st excited state.
Asked by patra04011965 | 28 Mar, 2020, 11:54: AM
potential energy U1 of ground state electron in Hydrogen atom is given by, 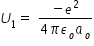
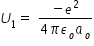
where e is charge of electron, εo is permittivity of free space and ao is Bohr radius
potential energy U2 of first excited state electron in Hydrogen atom is given by, 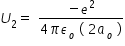
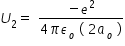
Above expression for potential energy is written by considering orbit radius for first excited state is 2ao
potential energy U of first excited state electron in Hydrogen atom with respect to ground state is given by,
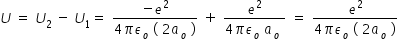
Speed of electron when it is in first excited state of hydrogen atom is obtained by
balancing electrostatic force with centripetal force
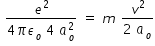 ....................(1)
....................(1)From above expression we get kinetic energy as , 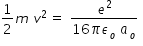
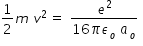
Hence total mechanical energy = Kinetic energy + Potential energy = 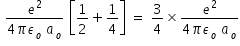
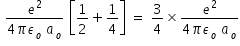
by substituting values e = 1.602 × 10-19 , Coulomb's constant 1/(4πεo ) = 9 × 109 N m2 C-2
and Bohr radius = 0.0529 nm
we get Total mechanical energy = 3.275 × 10-18 J
Answered by Thiyagarajan K | 28 Mar, 2020, 02:12: PM
Application Videos
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM