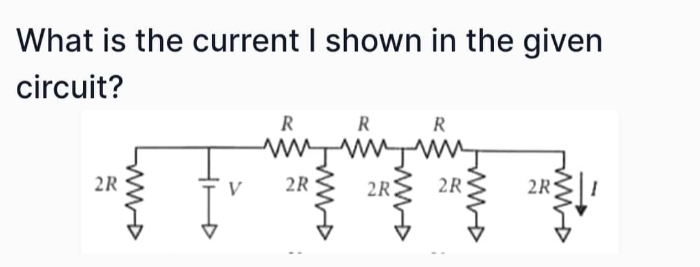
JEE Class main Answered
find partial pressue

Asked by ashutosharnold1998 | 25 Feb, 2020, 11:20: AM
number of oxygen mole = 20/32 = 0.625
number of CO mole = 2.8/28 = 0.1
mole fraction of oxygen = 0.625 / (0.625+0.1) = 0.625/0.725 = 25/29
mole fraction of CO = 0.1/0.725 = 4/29
Partial pressure of Oxygen = (25/29)×( 2 atm ) = ( 50/29 ) atm
Partial volume of oxygen = (25/29)×( 10 L ) = ( 250 / 29 ) L
Answered by Thiyagarajan K | 25 Feb, 2020, 11:55: PM
Application Videos
JEE main - Physics
Asked by arivaryakashyap | 23 Apr, 2024, 10:40: AM
JEE main - Physics
Asked by ratnadeep.dmr003 | 21 Apr, 2024, 11:06: PM
JEE main - Physics
Asked by ksahu8511 | 19 Apr, 2024, 11:55: AM
JEE main - Physics
Asked by mohammedimroz | 13 Apr, 2024, 09:48: PM
JEE main - Physics
Asked by medhamahesh007 | 02 Apr, 2024, 11:11: AM
JEE main - Physics
Asked by gundlasumathi93 | 31 Mar, 2024, 02:13: PM
JEE main - Physics
Asked by chhayasharma9494 | 31 Mar, 2024, 12:47: PM
JEE main - Physics
Asked by archithateja3 | 30 Mar, 2024, 10:23: PM
JEE main - Physics
Asked by Machinenineha | 27 Mar, 2024, 05:28: PM