CBSE Class 11-science Answered
Find out the emperical formula and molecular formula in organic compund contain 62.07 % Carbon,10% Hydrogen,and 14% Nitrogen. The molecular mass of compound is 114 gm/mole
Asked by Shreypanchalspiky | 06 Jul, 2018, 11:24: PM
The total composition is 100
Given:
Carbon=62.07
Hydrogen =10
Nitrogen =14
Oxygen =(100-86.07)
=13.93
|
Element |
% composition |
Atomic weight |
Ratio |
Simplest Ratio |
|
Carbon |
62.07 |
12 |
5.17 |
5.90= 6 |
|
Hydrogen |
10 |
1 |
10 |
11.4=11 |
|
Oxygen |
13.93 |
16 |
0.870 |
1 |
|
Nitrogen |
14 |
14 |
1 |
1.1=1 |
So the empirical formula is: C6H11NO
Empirical formula mass = 113
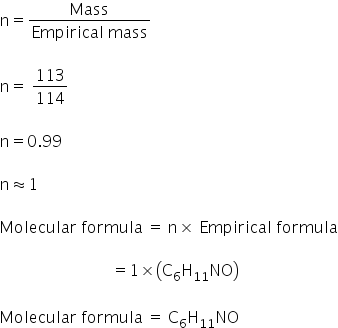
Empirical formula is C6H11NO
Molecular formula is C6H11NO
Answered by Varsha | 09 Jul, 2018, 08:30: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by drhimasingh | 22 May, 2020, 11:39: AM
CBSE 11-science - Chemistry
Asked by nareshrajpurohit43109 | 22 May, 2020, 11:18: AM
CBSE 11-science - Chemistry
Asked by d6knx7qmw1 | 15 May, 2020, 10:37: PM
CBSE 11-science - Chemistry
Asked by sahadipa1975 | 02 May, 2020, 08:53: AM
CBSE 11-science - Chemistry
Asked by abhishek19362771 | 08 Apr, 2020, 03:48: PM
CBSE 11-science - Chemistry
Asked by anilsolanki2060 | 22 Feb, 2020, 10:12: AM
CBSE 11-science - Chemistry
Asked by pujakurmi22 | 11 Nov, 2019, 10:59: PM
CBSE 11-science - Chemistry
Asked by jkatwara | 14 Oct, 2019, 12:21: PM
CBSE 11-science - Chemistry
Asked by vikas.kochhar6 | 30 Aug, 2019, 03:58: PM
CBSE 11-science - Chemistry
Asked by pb_ckt | 19 May, 2019, 11:56: PM