CBSE Class 9 Answered
Explain the process shown in the figure below:
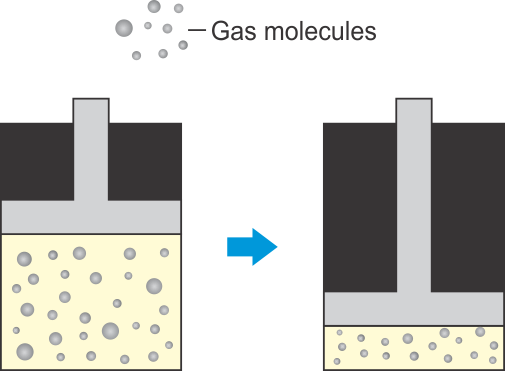

Asked by Topperlearning User | 05 Apr, 2016, 11:01: AM
There is a lot of space between the particles of a gas. We can reduce the spaces between the particles by compressing them by pushing in the piston in a cylinder. When a high pressure is applied to the piston, the particles get so close together that they start attracting each other sufficiently to form a liquid and the gas is liquefied.
Answered by | 05 Apr, 2016, 01:01: PM
Application Videos
Concept Videos
CBSE 9 - Chemistry
Asked by rubinapathan228 | 29 Jun, 2023, 05:45: PM
CBSE 9 - Chemistry
Asked by yforyt3672 | 09 Apr, 2023, 07:37: PM
CBSE 9 - Chemistry
Asked by pranavtamboli65.9 | 02 Aug, 2022, 08:12: PM
CBSE 9 - Chemistry
Asked by ritapriya126 | 20 Apr, 2022, 04:01: PM
CBSE 9 - Chemistry
Asked by renukaramaswamyr | 08 Sep, 2021, 12:32: PM
CBSE 9 - Chemistry
Asked by gpranithasri | 27 Jul, 2021, 01:23: PM
CBSE 9 - Chemistry
Asked by suhanivasi007 | 04 May, 2021, 05:52: PM
CBSE 9 - Chemistry
Asked by pk36229905 | 06 Apr, 2021, 07:38: AM
CBSE 9 - Chemistry
Asked by anubhavev | 30 Oct, 2020, 06:35: PM