NEET Class neet Answered
Equivalent weight is
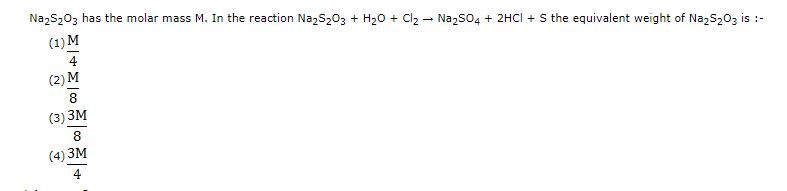
Asked by Mrinal | 20 May, 2019, 10:07: PM
Option (3) is correct.
Given:
Molecular weight of Na2S2O3 = M
The reaction is,
Na2S2O3 + H2O + Cl2 →Na2SO4 + 2HCl + S
The change in oxidation number for,
Na2S2O3 → Na2SO4 is
+2 → +6 = 4
For , Na2S2O3 → S
+2 → 0 = 2
We know,
Equivalent weight is,
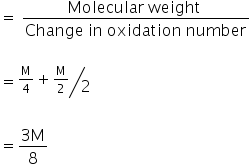
The equivalent weight of Na2S2O3 is 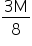 .
.
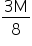 .
.
Answered by Varsha | 21 May, 2019, 10:45: AM
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vaka.aruna1979 | 23 Mar, 2024, 04:18: AM
NEET neet - Chemistry
Asked by fathimahusna23042004 | 03 Mar, 2024, 08:56: AM
NEET neet - Chemistry
Asked by drkeshavkhandagle | 18 Jan, 2024, 08:10: PM
NEET neet - Chemistry
Asked by yogitakumawat | 21 Dec, 2023, 10:31: PM






