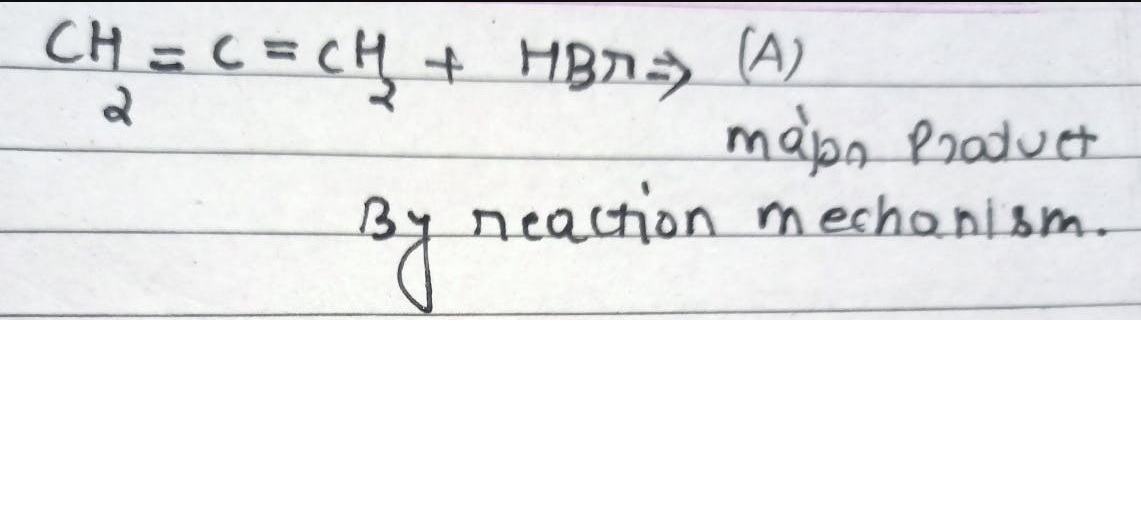CBSE Class 12-science Answered
Can you explain the reaction? And, why isn't it butan-2-ol ?
Asked by akshitaasia | 12 Mar, 2018, 01:46: PM
Don't get confused between the elimination reaction and the substitution reaction.
The difference between both reactions is medium.
Case 1: If the medium is aqueous then,
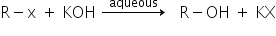
If the medium is aqueous then OH- will act as a nucleophile and the reaction will proceed via substitution to produce alcohol.
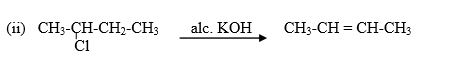
Case 2: If the medium is alcoholic then,
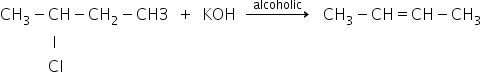
If the medium is alcoholic then OH- ion will act as a base and abstract proton then reaction proceeds via elimination.
That's why it will not give butan-2-ol it will only give elimination product that is the alkene
I hope now you will not confuse by both these reactions.
Answered by Ramandeep | 12 Mar, 2018, 03:57: PM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by surajbhanupatro44 | 07 Nov, 2023, 12:01: AM
CBSE 12-science - Chemistry
Asked by mayamishra9540500880 | 04 Jul, 2022, 07:11: PM
CBSE 12-science - Chemistry
Asked by harshaldpathak | 11 Jun, 2022, 05:37: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 01 Jan, 2021, 09:15: PM
CBSE 12-science - Chemistry
Asked by me.mirzainayat | 14 Nov, 2020, 07:31: AM
CBSE 12-science - Chemistry
Asked by Prachidewangan74 | 02 Oct, 2020, 03:02: PM
CBSE 12-science - Chemistry
Asked by sujithanathan119 | 01 Jun, 2020, 12:00: PM
CBSE 12-science - Chemistry
Asked by ng9045007209 | 21 May, 2020, 07:47: PM
CBSE 12-science - Chemistry
Asked by gangavaramouni | 26 Mar, 2020, 10:33: AM