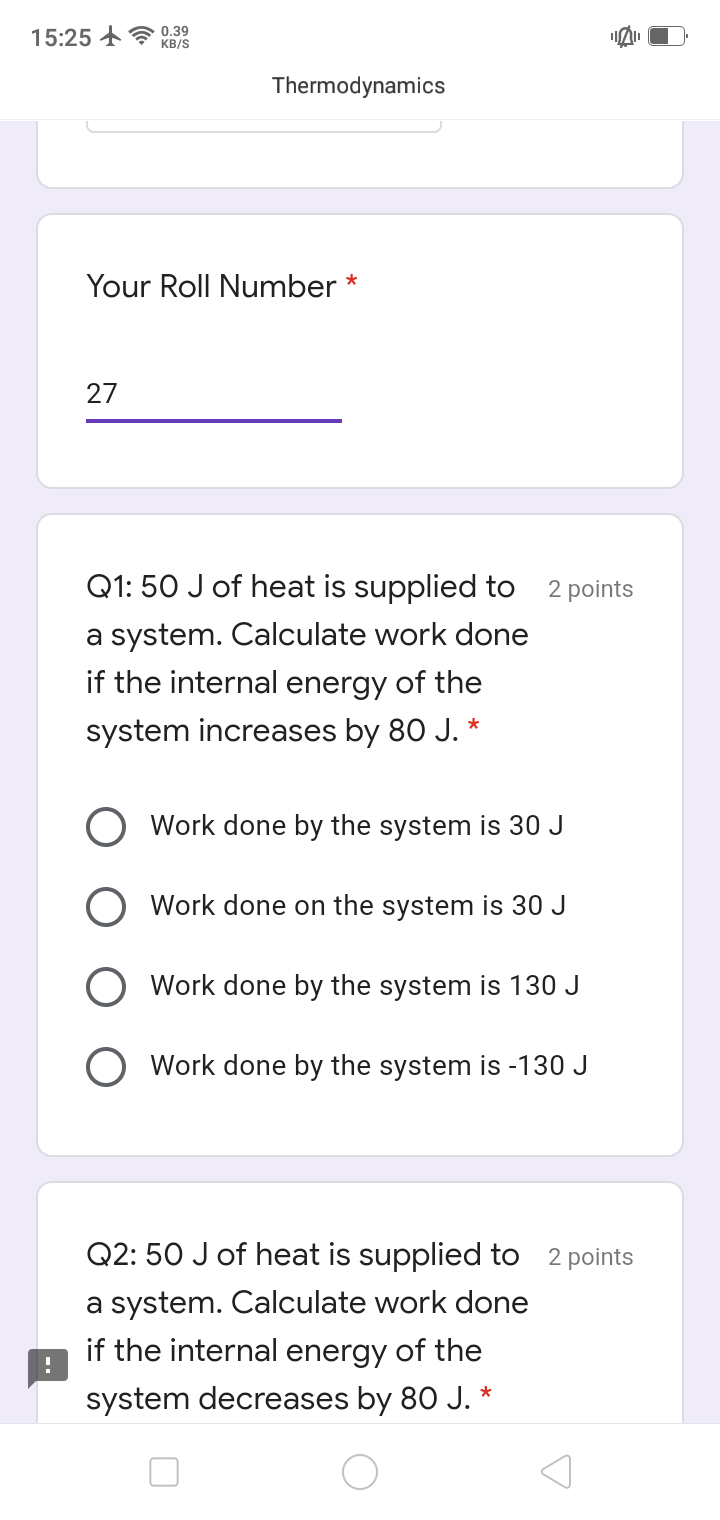
CBSE Class 11-science Answered
Calculate the enthalpy of decomposition of CaCO3 into CaO and CO2.The  of formation of CaCO3 is -288.5 k cal/mol, for CaO is -151.9 k cal/mol and for CO2.is -94.0 k cal/mol.
of formation of CaCO3 is -288.5 k cal/mol, for CaO is -151.9 k cal/mol and for CO2.is -94.0 k cal/mol.
 of formation of CaCO3 is -288.5 k cal/mol, for CaO is -151.9 k cal/mol and for CO2.is -94.0 k cal/mol.
of formation of CaCO3 is -288.5 k cal/mol, for CaO is -151.9 k cal/mol and for CO2.is -94.0 k cal/mol.
Asked by Topperlearning User | 15 Jun, 2016, 05:26: PM
The equation for the above reaction is
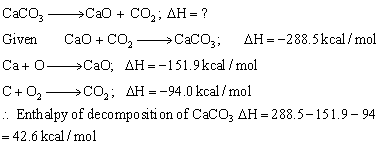
Answered by | 15 Jun, 2016, 07:26: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by kjay0981 | 13 Dec, 2020, 03:45: PM
CBSE 11-science - Chemistry
Asked by jain.pradeep | 14 Apr, 2019, 12:33: AM
CBSE 11-science - Chemistry
Asked by Atulcaald | 25 May, 2018, 12:31: AM
CBSE 11-science - Chemistry
Asked by gganga | 13 Apr, 2018, 06:34: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 13 Aug, 2014, 01:38: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 05:22: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 13 Aug, 2014, 02:12: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 15 Jun, 2016, 05:22: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 13 Aug, 2014, 02:32: PM


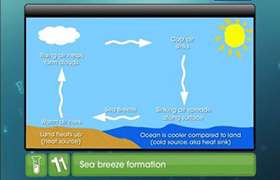

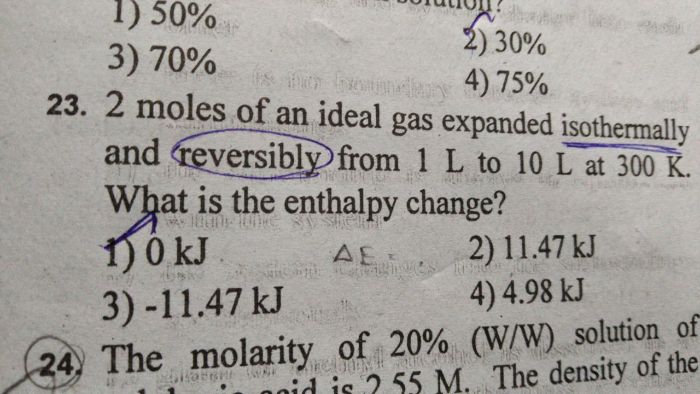

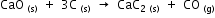 The heats of formation of CaO, CaC2 and CO are -151.6, -14.2 and -26.4 kcal respectively.
The heats of formation of CaO, CaC2 and CO are -151.6, -14.2 and -26.4 kcal respectively.