CBSE Class 12-science Answered
Calculate the amount of energy released during the 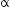 decay of
decay of 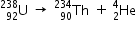 Given :
1.atomic mass of
Given :
1.atomic mass of 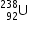 =238.05079u
2. atomic mass of
=238.05079u
2. atomic mass of 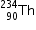 = 234.04363
3.atomic mass of
= 234.04363
3.atomic mass of 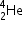 = 4.00260
lu = 931.5 MeV/c2.
Is this decay spontaneous? Give reason.
= 4.00260
lu = 931.5 MeV/c2.
Is this decay spontaneous? Give reason.
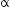 decay of
decay of 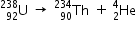 Given :
1.atomic mass of
Given :
1.atomic mass of 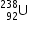 =238.05079u
2. atomic mass of
=238.05079u
2. atomic mass of 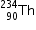 = 234.04363
3.atomic mass of
= 234.04363
3.atomic mass of 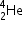 = 4.00260
lu = 931.5 MeV/c2.
Is this decay spontaneous? Give reason.
= 4.00260
lu = 931.5 MeV/c2.
Is this decay spontaneous? Give reason.
Asked by Topperlearning User | 09 Jul, 2014, 03:56: PM
The energy released in the 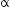 decay is
decay is
Q = 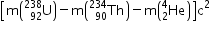
= [238.05079 - 234.04363 - 4.00260] x 931.5MeV
= 0.00456 x931.5 = 4.25MeV
As the Q - value is positive, the decay process is spontaneous.
Answered by | 09 Jul, 2014, 05:56: PM
Concept Videos
CBSE 12-science - Physics
Asked by arjunsah797 | 16 May, 2022, 02:17: PM
CBSE 12-science - Physics
Asked by merinlijo_20002 | 17 Jun, 2020, 10:05: AM
CBSE 12-science - Physics
Asked by rajubarman | 01 Dec, 2019, 10:03: AM
CBSE 12-science - Physics
Asked by alanpeter9611 | 23 Feb, 2019, 07:47: PM
CBSE 12-science - Physics
Asked by alanpeter9611 | 22 Feb, 2019, 11:24: PM
CBSE 12-science - Physics
Asked by sd2021667 | 04 Dec, 2018, 04:46: PM
CBSE 12-science - Physics
Asked by silladech | 16 Nov, 2018, 11:14: AM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:06: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:15: PM






 , find the number of nuclei present after 8 days?
, find the number of nuclei present after 8 days?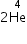 →
→  + ..............
+ ..............
