CBSE Class 11-science Answered
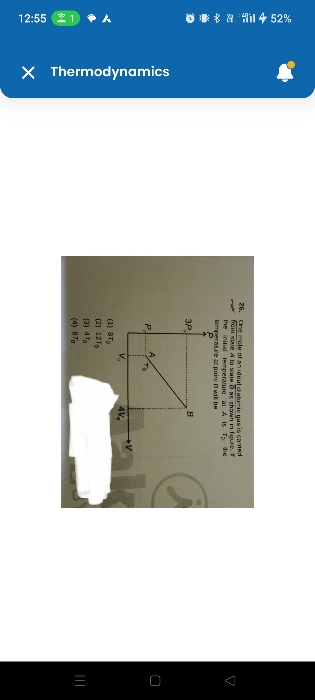
applying first law of thermodynamics obtain an adiabatic relation between pressure and volume.
Asked by ashwinikumar59 | 16 Feb, 2011, 07:13: AM
Dear student
A/C to 1st law of thermodynamics;
dQ= dU + PdV
For adiabatic process, dQ=0
So, dU + PdV=0
or, dU = -PdV
When the expansion takes place, volume of the gas increases i.e. dV = +ve, hence dU = -ve
;
i.e. internal energy of the gas decreases and hence the temperature falls.
If the gas is compressed, volume of the gas decreases, i.e. dV = -ve, hence dU = +ve; i.e.
internal energy of the gas increase and hencew its temperature increase.
Hope this clarifies your doubt.
Regards
Team
Topperlearning
Answered by | 16 Feb, 2011, 09:40: AM
Concept Videos
CBSE 11-science - Physics
Asked by shubham23302007 | 23 Jan, 2024, 10:24: PM
CBSE 11-science - Physics
Asked by s3043632 | 22 Jan, 2023, 06:45: PM
CBSE 11-science - Physics
Asked by juzarsiddhapurwala | 07 Mar, 2021, 10:08: PM
CBSE 11-science - Physics
Asked by bbabichowdary | 02 Sep, 2020, 01:25: PM
CBSE 11-science - Physics
Asked by rajualpine16 | 19 Aug, 2020, 07:42: AM
CBSE 11-science - Physics
Asked by hazim.mohaamad2003 | 02 Mar, 2019, 12:10: PM
CBSE 11-science - Physics
Asked by abhishekmishra1031 | 12 Feb, 2019, 03:10: PM
CBSE 11-science - Physics
Asked by govtsecschoolnayaganv051 | 14 Dec, 2018, 07:19: PM
CBSE 11-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM