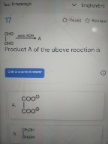
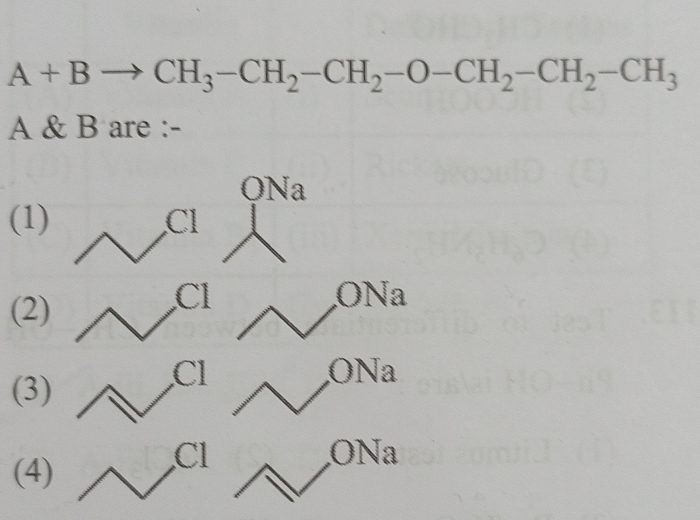
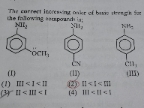
NEET Class neet Answered
An aqueous sol is 1.00m molal in KI Which change will cause vapour pressure of the sol to increase?
(a) addition of NaCl (b) addition of 1.00m molal KI (c) addition of water
Pls provide reason for why and (c) is correct !!
And also pls mention that why option (b) is not correct !!
And the reason I have assumed for which option (a) is wrong is that - NaCl is non volatile and so adding it will decrease vapour pressure so am I right ??
Asked by charvipareek057 | 19 Apr, 2019, 08:32: AM

Answered by Sumit Chakrapani | 20 Apr, 2019, 02:00: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by biswassayan8464 | 21 Apr, 2024, 11:30: AM
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM
NEET neet - Chemistry
Asked by ankuruthanuriya | 03 Apr, 2024, 10:56: PM