CBSE Class 11-science Answered
Air contains 20% oxygen by volume how much volume of air will be required for combustion of 100 cc acetylene
Why we use at last as 100/20*250
Asked by gchoudhary2697 | 04 Aug, 2019, 09:55: AM
2C2H2 + 5O2 → 4CO2 + 2H2O
2 moles of C2H2 react with 5 moles of O2
1 mole of C2H2 = 22.44 cc
100 cc of C2H2
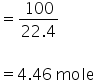
2 moles of C2H2 react with 5 moles of O2
4.346 mole of C2H2 will react with
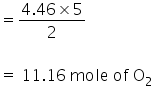
= 11.16×22.4
= 250 cc of O2
Since air contains 20% of oxygen by volume, the amount of air needed to react with 100 cc of C2H2
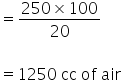
Answered by Varsha | 04 Aug, 2019, 05:52: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by jayag1298 | 08 Apr, 2024, 03:14: PM
CBSE 11-science - Chemistry
Asked by omniscientnjf2021 | 07 Apr, 2024, 10:18: PM
CBSE 11-science - Chemistry
Asked by hcnainwal | 15 Jun, 2023, 10:39: AM
CBSE 11-science - Chemistry
Asked by Jprmumal29 | 18 Dec, 2022, 09:48: PM
CBSE 11-science - Chemistry
Asked by mallikarjunasangi28 | 22 Jul, 2022, 07:57: PM
CBSE 11-science - Chemistry
Asked by vedwatisharma79 | 10 Jun, 2022, 05:27: PM
CBSE 11-science - Chemistry
Asked by thathvakunjusree | 10 Dec, 2021, 06:46: AM
CBSE 11-science - Chemistry
Asked by udheshraddha2004 | 28 Oct, 2021, 09:37: PM
CBSE 11-science - Chemistry
Asked by arunparewa2000 | 27 Oct, 2021, 06:59: PM
CBSE 11-science - Chemistry
Asked by arttameher038 | 23 Aug, 2021, 07:06: AM







