CBSE Class 10 Answered
A silvery white metal X reacts with water at room temperature to produce a water soluble compound Y and a colourless gas Z. The reactions is highly exothermic and the Z catches fire immediately during the reaction. The solution of Y in water on reacting with stoichiometric amount of dilute solution of hydrochloric acid gives a solution of pH = 7.0. Name the compounds X,Y and Z ?
Asked by rushabh123 | 14 Mar, 2019, 10:52: AM
A silvery-white metal X reacts with water at room temperature to produce a water-soluble compound Y and a colourless gas Z.
Sodium, a silvery-white metal reacts with water to produce sodium hydroxide and hydrogen gas is evolved.
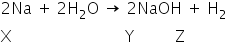
Answered by Varsha | 14 Mar, 2019, 04:37: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by ritik9897022 | 05 Feb, 2024, 09:42: PM
CBSE 10 - Chemistry
Asked by shubham.sharma80634 | 05 Oct, 2021, 09:18: AM
CBSE 10 - Chemistry
Asked by virkarman36 | 08 Aug, 2021, 09:24: AM
CBSE 10 - Chemistry
Asked by dnupadhyay214 | 13 Mar, 2021, 12:01: PM
CBSE 10 - Chemistry
Asked by Vishavjet567 | 31 Oct, 2020, 10:52: AM
CBSE 10 - Chemistry
Asked by aryanluniwal1516 | 12 Sep, 2020, 11:43: AM
CBSE 10 - Chemistry
Asked by broprint18 | 07 Jun, 2020, 04:16: PM
CBSE 10 - Chemistry
Asked by prakharsingh167 | 25 May, 2020, 10:20: PM
CBSE 10 - Chemistry
Asked by sonaliagarwal172 | 16 May, 2020, 10:16: AM