CBSE Class 12-science Answered
A radioactive sample contains 2.2 mg of pure  which has half life period of 1224 seconds. Calculate
(i) the number of atoms present initially.
(ii) the activity when 5μg of the sample will be left.
which has half life period of 1224 seconds. Calculate
(i) the number of atoms present initially.
(ii) the activity when 5μg of the sample will be left.
 which has half life period of 1224 seconds. Calculate
(i) the number of atoms present initially.
(ii) the activity when 5μg of the sample will be left.
which has half life period of 1224 seconds. Calculate
(i) the number of atoms present initially.
(ii) the activity when 5μg of the sample will be left.
Asked by Topperlearning User | 09 Jul, 2014, 06:46: PM
(i) By definition 11g of carbon  contains NA = 6.023 x 1023 atoms
contains NA = 6.023 x 1023 atoms
number of atoms in 2.2 mg = 2.2 x 10-3g of  will be
will be
= 
(ii) Activity A = λN
Half life T1/2 =
Activity A = 
Where N = Number of atoms in 5μg (= 5 x 10-6g) which is equal to


Answered by | 09 Jul, 2014, 08:46: PM
Concept Videos
CBSE 12-science - Physics
Asked by arjunsah797 | 16 May, 2022, 02:17: PM
CBSE 12-science - Physics
Asked by merinlijo_20002 | 17 Jun, 2020, 10:05: AM
CBSE 12-science - Physics
Asked by rajubarman | 01 Dec, 2019, 10:03: AM
CBSE 12-science - Physics
Asked by alanpeter9611 | 23 Feb, 2019, 07:47: PM
CBSE 12-science - Physics
Asked by alanpeter9611 | 22 Feb, 2019, 11:24: PM
CBSE 12-science - Physics
Asked by sd2021667 | 04 Dec, 2018, 04:46: PM
CBSE 12-science - Physics
Asked by silladech | 16 Nov, 2018, 11:14: AM
CBSE 12-science - Physics
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:06: PM
CBSE 12-science - Physics
Asked by Topperlearning User | 09 Jul, 2014, 04:15: PM






 , find the number of nuclei present after 8 days?
, find the number of nuclei present after 8 days?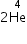 →
→  + ..............
+ ..............
