NEET Class neet Answered
A→→C(Half life=1 minute), B→→C(Half life=2minutes). At t=0, concentration of A was 4N。 whereas concentration of B was N。 at t=0. Find- (i) the concentration of C at the instance when concentration of A and B becomes equal. Also find(ii) the concentration of C at the instance when activity of A becomes equal to activity of B.
Asked by patra04011965 | 30 Mar, 2020, 08:02: PM
Concentration of A at time t , 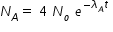 ........................ (1)
........................ (1)
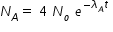 ........................ (1)
........................ (1)Concentration of B at time t , 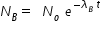 .........................( 2 )
.........................( 2 )
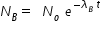 .........................( 2 )
.........................( 2 )where λA is decay constant of A and it is given by, λA = ln2 min-1
where λB is decay constant of B and it is given by, λB = (1/2) ln2 min-1
Time t for getting equal concentration of A and B is obtained by equating above equation and solving for time t
If NA = NB , then we have , 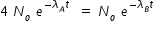
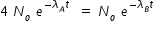
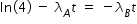
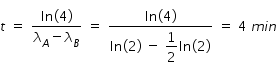
Hence concentration of C at t = 4 min given by,
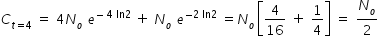
----------------------------------------------------------
Activity of A , 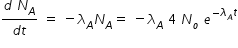
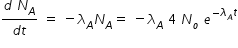
Activity of B , 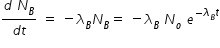
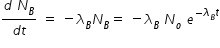
If both activities are equal, then we have,
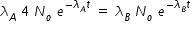
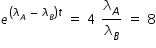
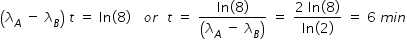
Hence concentration of C at t = 6 min given by,
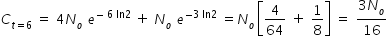
Answered by Thiyagarajan K | 30 Mar, 2020, 10:40: PM
Application Videos
NEET neet - Physics
Asked by shatakshibhatt9 | 20 Apr, 2024, 07:52: PM
NEET neet - Physics
Asked by praveenpriya000079 | 18 Apr, 2024, 07:24: AM
NEET neet - Physics
Asked by gouranshi84 | 17 Apr, 2024, 05:23: PM
NEET neet - Physics
Asked by sojusvi | 17 Apr, 2024, 01:12: PM