CBSE Class 12-science Answered
Please answer the following question in detail, specifically the query that is mentioned in the image.

Asked by Balbir | 22 Jun, 2018, 02:16: PM
Above reactions are exceptional reactions
These reactions of salt of oxyacids are given by Iodine only because Iodine is more electropositive than Chlorine and Bromine.
The main answer key to these reactions is ClO3- is a strong oxidizing agent than IO3-, hence Cl or Br is replaced by Iodine. As above said, these two are exceptional reactions.
Only following two reactions of oxyacids occur as follows:
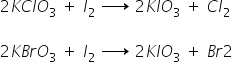
The above reactions may be verified from half-cell potentials,
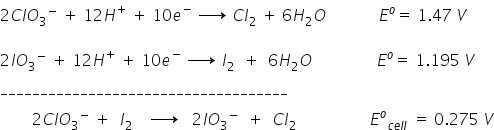
Now, Hope you understand these exceptional reactions.
Hence the option D is the correct answer.
Answered by Ramandeep | 23 Jun, 2018, 07:48: PM
Concept Videos
CBSE 12-science - Chemistry
Asked by nonuhasan2 | 20 Nov, 2021, 12:05: PM
CBSE 12-science - Chemistry
Asked by ecstayatra | 07 Apr, 2020, 08:53: PM
CBSE 12-science - Chemistry
Asked by dwivedipradeep093 | 02 May, 2019, 09:21: PM
CBSE 12-science - Chemistry
Asked by prashantrairohit | 10 Feb, 2019, 09:25: AM
CBSE 12-science - Chemistry
Asked by lekhakarthikeyan | 28 Aug, 2018, 03:49: AM
CBSE 12-science - Chemistry
Asked by Balbir | 22 Jun, 2018, 02:16: PM
CBSE 12-science - Chemistry
Asked by Balbir | 19 Jun, 2018, 06:33: PM
CBSE 12-science - Chemistry
Asked by my3shyll | 18 Apr, 2018, 06:46: PM
CBSE 12-science - Chemistry
Asked by scholarreema | 14 Mar, 2018, 12:22: PM
CBSE 12-science - Chemistry
Asked by Topperlearning User | 04 Jun, 2014, 01:23: PM





