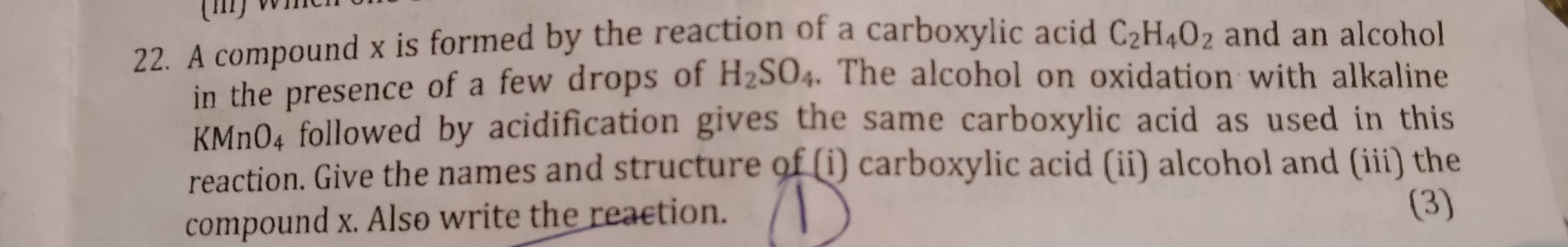CBSE Class 10 Answered
Explain the cleansing action of soap
Asked by abhisar | 17 Feb, 2018, 06:04: PM
When soap is dissolved in water, its hydrophobic ends attach themselves to dirt and remove it from the cloth.
First, the molecules of soap arrange themselves in micelle formation and trap the dirt at the centre of the cluster.
These micelles remain suspended in water like particles in a colloidal solution.
The various micelles present in water do not come together to form a precipitate as each micelle repels the other because of the ion-ion repulsion.
Thus, the dust particles remain trapped in micelles (which remain suspended) and are easily rinsed away with water.
Hence, soap micelles remove dirt by dissolving it in water.
Answered by Varsha | 17 Feb, 2018, 06:13: PM
Application Videos
Concept Videos
CBSE 10 - Chemistry
Asked by sneh | 27 Mar, 2020, 10:11: AM
CBSE 10 - Chemistry
Asked by pranjaliinamdar2004 | 29 Feb, 2020, 07:20: PM
CBSE 10 - Chemistry
Asked by sweetykhatri99254 | 27 Feb, 2020, 03:40: PM
CBSE 10 - Chemistry
Asked by priyanshiishu | 30 Jan, 2020, 10:39: AM
CBSE 10 - Chemistry
Asked by kamalnayansingh7 | 13 Jan, 2020, 08:35: AM
CBSE 10 - Chemistry
Asked by Deepak | 22 Dec, 2019, 11:20: PM
CBSE 10 - Chemistry
Asked by ritikraghuwanshi6986 | 16 Dec, 2019, 08:42: PM
CBSE 10 - Chemistry
Asked by vedantsagrawal23 | 05 Dec, 2019, 08:34: AM
CBSE 10 - Chemistry
Asked by aryasaxena2003 | 25 Jul, 2019, 05:35: PM
CBSE 10 - Chemistry
Asked by rushabhjain.avv | 21 Mar, 2019, 10:07: PM