CBSE Class 11-science Answered
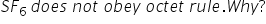
Asked by surajsubedi14 | 03 Mar, 2015, 10:29: AM
SF6 is a covalent bonded compound and has expanded octet.
In SF6 molecule, the central sulphur atom has twelve electrons, six electrons of its own and six electrons from six F atoms.
Therefore , SF6 does not obey octet rule.
Answered by Vaibhav Chavan | 03 Mar, 2015, 11:23: AM
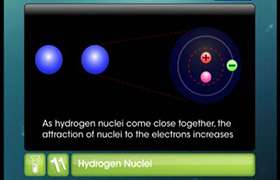
Concept Videos
CBSE 11-science - Chemistry
Asked by visalvinod06 | 23 Jun, 2022, 07:39: AM
CBSE 11-science - Chemistry
Asked by bhagwatkrutika6 | 22 Jun, 2022, 09:53: PM
CBSE 11-science - Chemistry
Asked by naveenbahuguna05 | 11 Dec, 2021, 03:19: PM
CBSE 11-science - Chemistry
Asked by akhileshpandeypandey456 | 12 Aug, 2021, 11:09: PM
CBSE 11-science - Chemistry
Asked by toshan.dandi5 | 24 Feb, 2021, 09:24: PM
CBSE 11-science - Chemistry
Asked by patilanil3766 | 01 Jan, 2021, 10:28: PM
CBSE 11-science - Chemistry
Asked by anjalikri2201 | 08 Aug, 2020, 11:22: AM
CBSE 11-science - Chemistry
Asked by khushipatel2124 | 07 Jul, 2020, 04:23: PM
CBSE 11-science - Chemistry
Asked by saurabhchaddha4205 | 03 Jun, 2020, 04:22: PM
CBSE 11-science - Chemistry
Asked by utkarshnikam85.tl | 06 Apr, 2020, 12:13: PM