NEET Class neet Answered
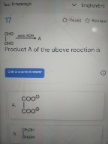
પાણીના એક નમૂનાનું ca+આયનની સંદ્રતા 0.0002M છે તો તે દ્રાવણમાં ca+ ની સન્દ્રતા વજન કદ થી ppm મા કેટલી થાય
Asked by vyompatel447 | 03 Jun, 2023, 03:35: AM
Dear Student,
Question statement is: If the concentration of Ca+ ions in a water sample is 0.0002M. then what is the concentration of Ca+ ions in the solution in ppm (parts per million)?
Solution:
1 ppm = 1 mg / L
Molar mass of Ca+ ion = 40 g/mol
Given, Molar concemtration = 0.0002 M
Mass of Ca+ ions in litre = (40 g /mol) × 0.0002 M
= 0.008 g / L = 8 mg / L = 8 ppm of Ca+ ion solution.
Answered by | 03 Jun, 2023, 01:22: PM
Application Videos
Concept Videos
NEET neet - Chemistry
Asked by mahendar160786 | 16 Apr, 2024, 09:23: PM
NEET neet - Chemistry
Asked by raomayankup83 | 15 Apr, 2024, 07:46: PM
NEET neet - Chemistry
Asked by muskannawab11 | 14 Apr, 2024, 03:13: PM
NEET neet - Chemistry
Asked by 8239682116rahul | 10 Apr, 2024, 01:48: PM
NEET neet - Chemistry
Asked by tarasingrathod63 | 07 Apr, 2024, 01:07: PM
NEET neet - Chemistry
Asked by fathimahusna6122 | 05 Apr, 2024, 10:25: AM
NEET neet - Chemistry
Asked by vasantagomasi23 | 05 Apr, 2024, 08:35: AM
NEET neet - Chemistry
Asked by ankuruthanuriya | 03 Apr, 2024, 10:56: PM
NEET neet - Chemistry
Asked by ghousiakaneez | 03 Apr, 2024, 12:55: PM