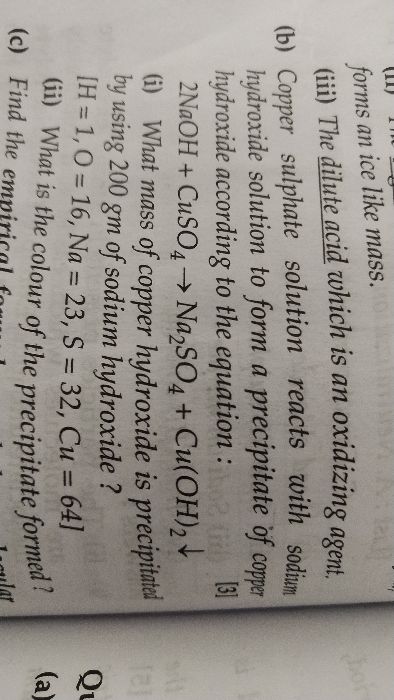ICSE Class 10 Answered
Zn+4HNO3 gives Zn[NO3]2+2H2O+2NO2 ANSWER THE FOLLOWING QUESTION : 1 The volume of NO2 formed at 19.5 g of Zn 2 The no of moles of HNO3 taking part in the reaction 3 The no of molecules of H2O formed 4 what mass of HNO3 is required to produce 145.6L of NO2 at S.T.P
Asked by nibeditamohapatro.publicschool | 15 Aug, 2018, 12:16: AM
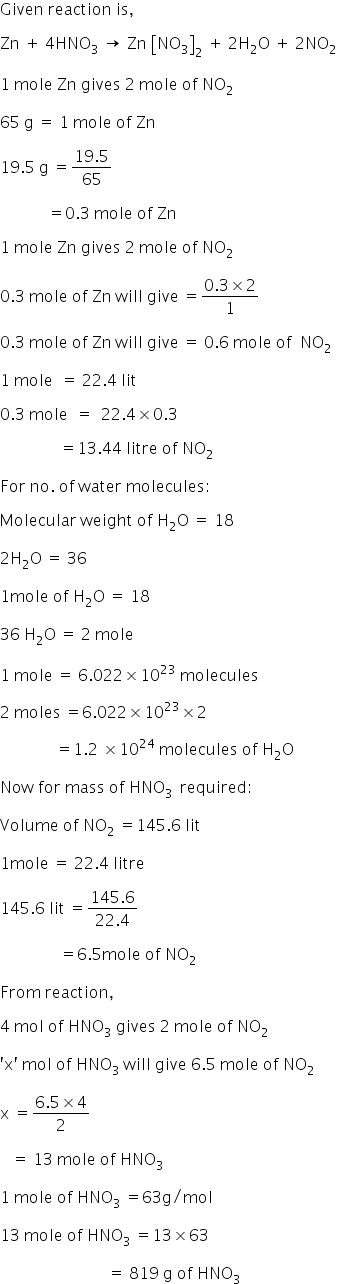
Answered by Varsha | 16 Aug, 2018, 02:52: PM
Application Videos
Concept Videos
ICSE 10 - Chemistry
Asked by jrvedant208 | 05 Feb, 2024, 10:37: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 10 Jul, 2022, 10:13: PM
ICSE 10 - Chemistry
Asked by rashikulkarni28 | 25 Jun, 2022, 10:24: PM
ICSE 10 - Chemistry
Asked by palshivom72 | 14 Jul, 2020, 07:56: PM
ICSE 10 - Chemistry
Asked by jhabijay01 | 27 May, 2020, 12:20: PM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:53: AM
ICSE 10 - Chemistry
Asked by aashimegh | 04 Sep, 2019, 08:37: AM
ICSE 10 - Chemistry
Asked by aashimegh | 28 Aug, 2019, 05:25: PM