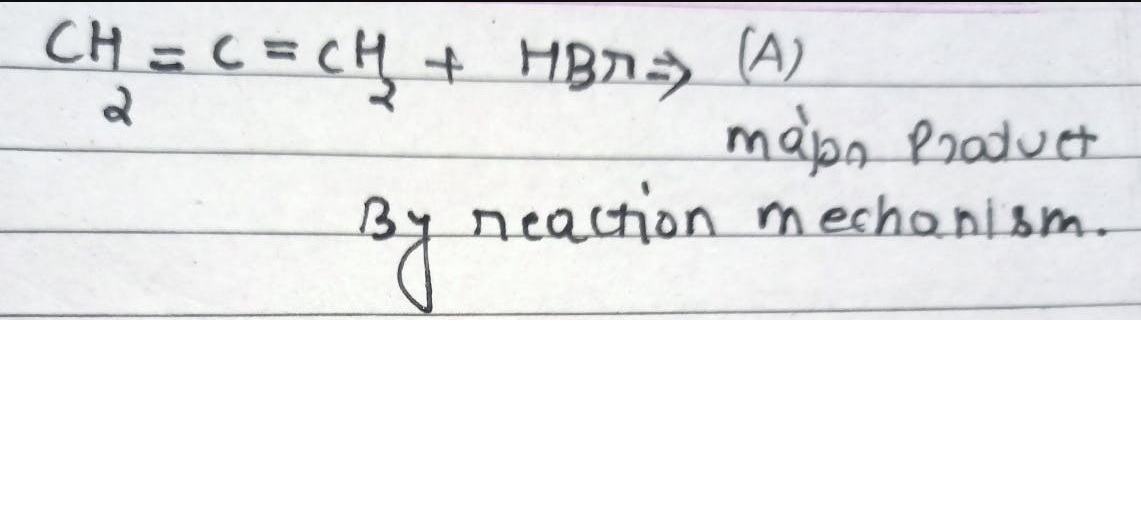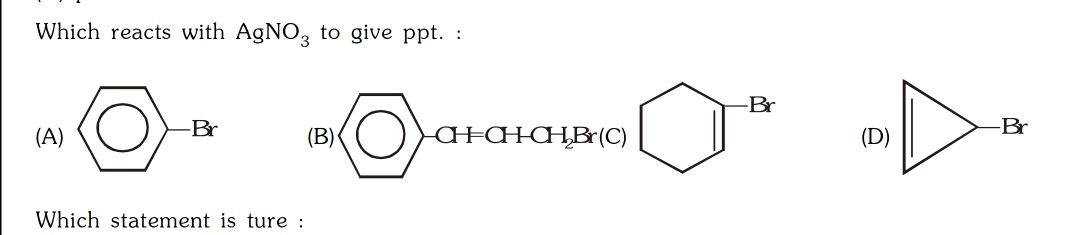CBSE Class 12-science Answered
word problem??
Asked by | 29 Feb, 2008, 05:09: AM
an optically active compound 'A' C4H9Br, on reduction, gives optically inactive compound 'B'. 'A' on reaction with KOH(alc.) gives 'C' which on reaction with HBr gives 'A' back. identify A,B and C and write the chemical reactions involved?
A is CH3CH2CHBrCH3 will be optically active with the chiral carbon atom.
B is CH3CH2CH2CH3
C isCH3CH=CHCH3 elimination will be by satzeffs rule.
Answered by | 20 Dec, 2017, 04:22: PM
Application Videos
Concept Videos
CBSE 12-science - Chemistry
Asked by surajbhanupatro44 | 07 Nov, 2023, 12:01: AM
CBSE 12-science - Chemistry
Asked by mayamishra9540500880 | 04 Jul, 2022, 07:11: PM
CBSE 12-science - Chemistry
Asked by harshaldpathak | 11 Jun, 2022, 05:37: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 01 Jan, 2021, 09:15: PM
CBSE 12-science - Chemistry
Asked by me.mirzainayat | 14 Nov, 2020, 07:31: AM
CBSE 12-science - Chemistry
Asked by Prachidewangan74 | 02 Oct, 2020, 03:02: PM
CBSE 12-science - Chemistry
Asked by sujithanathan119 | 01 Jun, 2020, 12:00: PM
CBSE 12-science - Chemistry
Asked by ng9045007209 | 21 May, 2020, 07:47: PM
CBSE 12-science - Chemistry
Asked by gangavaramouni | 26 Mar, 2020, 10:33: AM