CBSE Class 11-science Answered
Why this type of structure of ozone is not possible?
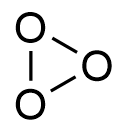
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 12:34: PM
In case of formation of any compound, stability is an important factor.
Because only if the compound is stable then it will exist.
For cyclic ozone, if this cyclic structure will exist then it has to face angle strains, lone pair-bond pair repulsion, electronic repulsion in a small three remembered ring (angular strain). In such abnormal conditions, and instability, ozone ring not generally found in nature.
Ozone is found in bend structure,
Answered by Ramandeep | 03 Jan, 2019, 02:45: PM
Concept Videos
CBSE 11-science - Chemistry
Asked by ABHILASHA | 08 Sep, 2019, 06:46: PM
CBSE 11-science - Chemistry
Asked by vishakhachandan026 | 12 Jun, 2019, 09:20: AM
CBSE 11-science - Chemistry
Asked by pb_ckt | 28 Apr, 2019, 01:40: PM
CBSE 11-science - Chemistry
Asked by satya785583 | 16 Mar, 2019, 09:18: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 01:09: PM
CBSE 11-science - Chemistry
Asked by govtsecschoolnayaganv051 | 03 Jan, 2019, 12:34: PM
CBSE 11-science - Chemistry
Asked by anushkamoudgil123 | 02 Nov, 2018, 05:47: AM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 04 Jul, 2018, 09:11: PM
CBSE 11-science - Chemistry
Asked by arunavamitra50 | 04 Jul, 2018, 09:10: PM
CBSE 11-science - Chemistry
Asked by Topperlearning User | 31 May, 2016, 01:10: PM






