CBSE Class 12-science Answered
Why is copper always found in a combined state despite occupying a lower position in the reactivity series?
Asked by lekhakarthikeyan | 06 Jul, 2018, 07:44: PM
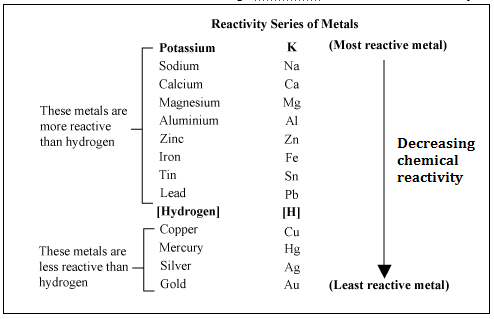
In the above reactivity series, Cu is less reactive and Au (Gold) is the least reactive.
Being less reactive Copper can react but the process goes slowly, hence copper found in nature in a combined form as copper pyrites CuFeS2.
Also, less reactive mercury as Cinnabar HgS, and silver as Argentite Ag2S are found in nature.
Only gold which is least reactive is found in nature in its native form or a free state Au.
Answered by Ramandeep | 12 Jul, 2018, 11:23: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by utsavs858 | 05 Mar, 2021, 12:36: AM
CBSE 12-science - Chemistry
Asked by akhilspillai2005 | 08 Nov, 2020, 03:17: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:39: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:38: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:38: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 28 Feb, 2020, 11:37: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 27 Feb, 2020, 02:48: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 27 Feb, 2020, 02:48: PM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 26 Feb, 2020, 09:59: AM
CBSE 12-science - Chemistry
Asked by sayleeashelar | 22 May, 2019, 11:32: AM