CBSE Class 12-science Answered
Why does bromination of aniline, even under very mild conditiongive 2,4,6-tribromoaniline instantaneoqsly.
Asked by BAKHORI AZAD | 10 Nov, 2010, 10:03: PM
Dear Student
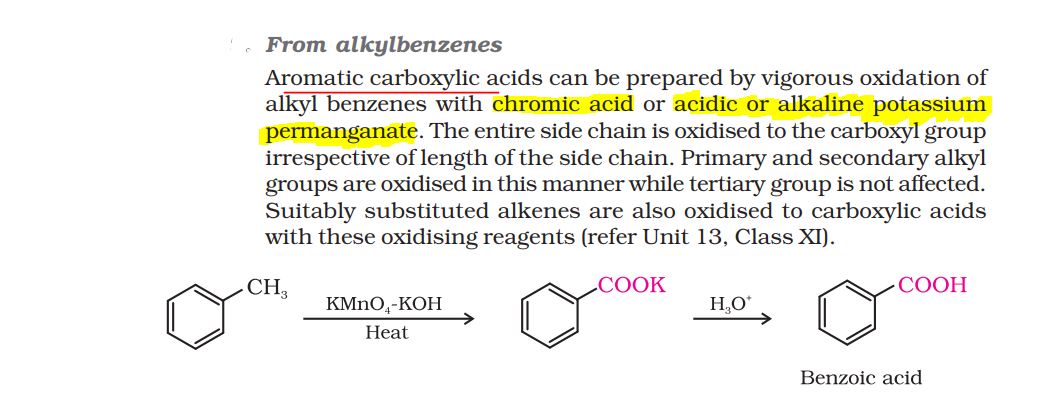
Aniline is very active towards bromination, an electrophilic aromatic substitution reaction.
Because the NH2 group of aniline is electron donating, which makes the aromatic ring that it's attached to electron rich. This is a perfect environment for the addition of an electrophilic reagent like bromine.
We hope that clarifies your query.
Regards
Team
Topperlearning
Answered by | 11 Nov, 2010, 09:50: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by ukg8612 | 15 Apr, 2024, 07:36: PM
CBSE 12-science - Chemistry
Asked by ajayarchi | 08 Feb, 2024, 03:43: AM
CBSE 12-science - Chemistry
Asked by pallasriramulu9 | 24 Dec, 2023, 06:05: AM
CBSE 12-science - Chemistry
Asked by bsaheliya | 22 Dec, 2023, 09:53: PM
CBSE 12-science - Chemistry
Asked by ygarg8323 | 18 Apr, 2022, 12:47: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 30 Jun, 2021, 04:52: PM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 29 Jun, 2021, 08:36: AM
CBSE 12-science - Chemistry
Asked by kaziryan.05 | 28 Jun, 2021, 02:34: PM
CBSE 12-science - Chemistry
Asked by saimerala007 | 22 May, 2021, 02:08: PM
CBSE 12-science - Chemistry
Asked by amitkumar.cis | 31 Dec, 2020, 10:45: AM