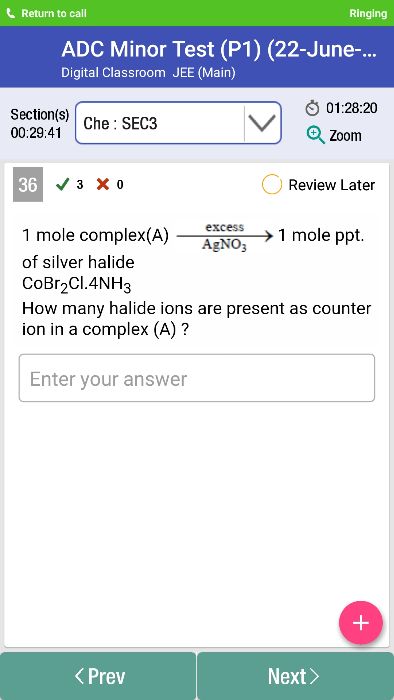
CBSE Class 12-science Answered
why cyanide ions are not considered as ambidentated ligand though it can act as an isocyadide ion?
Asked by vasturushi | 07 Nov, 2017, 07:33: PM
Ambidentated ligand: Ligands those have two different donor atoms
For example NO2, SCN, CN
[Cu(CN)4 ]3- In this coordination compound, CN is the ligand, CN can be attached to central metal atom i.e. Cu through C (Cu → CN) and through N (Cu→ NC)
This is possible because here, in this case, the transfer of charge is possible, the -ve charge can move between C and N.
Answered by Ramandeep | 08 Nov, 2017, 10:35: AM
Concept Videos
CBSE 12-science - Chemistry
Asked by prathyushagn1 | 09 Dec, 2020, 08:12: AM
CBSE 12-science - Chemistry
Asked by ABHILASHA | 31 Aug, 2020, 08:24: PM
CBSE 12-science - Chemistry
Asked by sha.bijoy17 | 07 Aug, 2020, 11:55: AM
CBSE 12-science - Chemistry
Asked by Shambhuhd79 | 22 Jun, 2020, 11:09: AM
CBSE 12-science - Chemistry
Asked by jain.pradeep | 19 Feb, 2020, 09:20: AM
CBSE 12-science - Chemistry
Asked by smit230503 | 04 Feb, 2020, 08:56: PM
CBSE 12-science - Chemistry
Asked by monishadubey202 | 08 Jan, 2020, 03:42: PM
CBSE 12-science - Chemistry
Asked by Chakshu29saini | 17 Sep, 2019, 06:19: PM
CBSE 12-science - Chemistry
Asked by bjayanta | 24 Mar, 2019, 08:56: PM
CBSE 12-science - Chemistry
Asked by himanshuneb | 28 Jan, 2019, 10:33: PM